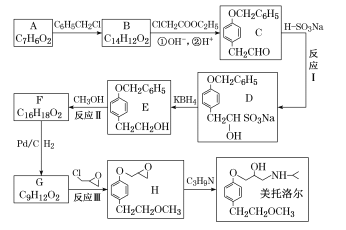
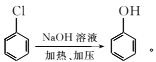
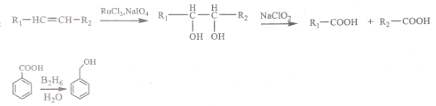
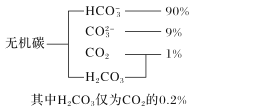
��Ŀ����
����Ŀ�������£���ijһԪ�� HA �� NaOH ��Һ�������ϣ�������Һ��Ũ�Ⱥͻ�Ϻ�������Һ�� pH �����
ʵ���� | HA���ʵ���Ũ��/(molL-1) | NaOH���ʵ���Ũ��/( molL-1) | �����Һ��pH |
�� | 0.1 | 0.1 | pH��9 |
�� | c | 0.2 | pH��7 |
�� | 0.2 | 0.2 | pH��a |
��ش�
��1�������ӷ���ʽ��ʾ��������Һ�Լ��Ե�ԭ����_____��
��2��������c_____0.2(���������������������ͬ)�������� a_____9��
��3�������£�HA �ĵ���ƽ�ⳣ����ֵΪ_____��
��4��������ʵ��������� pH��11 �� NaOH ��Һ�� pH��3 �� HA ��Һ�������ϣ����û����Һ����������Ũ���ɴ�С��˳��Ϊ_____��
���𰸡�A����H2OHA��OH�� �� �� 5��10-6 c(A)��c(Na+)��c(H+)��c(OH)
��������
(1)����ǡ�÷�Ӧ����NaA����Һ�ʼ��ԣ�˵��NaA��ǿ�������Σ��������ˮ�����Һ�ʼ��ԣ�ˮ�����ӷ���ʽΪA+H2OHA+OH��
(2)����Ϊǿ�ᣬ�������Ũ�Ȼ��ʱpH=7����HAΪ���ᣬ�������Ũ�Ȼ����Һ��pH����7����Ϊ��֤pH=7��Ӧʹ��Ũ�ȴ���0.2mol/L������ͼ��0.1mol/Lʱ��pH��9������ͼ��0.2mol/Lʱ���õ���ǿ�������αȢ��е��ε�Ũ�ȴʼ���Խǿ��a��9��
(3) HA�������ᣬ����ͼ��0.1mol/Lʱ����Ϻ����ҺΪNaA��pH��9��c(H+)=10-9mol/L��c(OH-)=![]() mol/L=10-5���跢��ˮ���A-��Ũ��Ϊx��
mol/L=10-5���跢��ˮ���A-��Ũ��Ϊx��
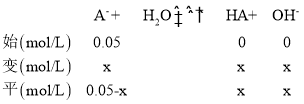
Kh=![]() ����Һ������������������ˮ����x= c(OH-)= 10-5�����Kh=2��10-9����Ka=
����Һ������������������ˮ����x= c(OH-)= 10-5�����Kh=2��10-9����Ka=![]() =
=![]() =5��10-6 ��
=5��10-6 ��
(4)pH��3 �� HA ��Ũ�ȴ���pH��11 �� NaOH ��Һ��Ũ�ȣ��������Ϻ���ʣ�࣬��Һ�����ԣ���c(H+)��c(OH)�����ݵ���غ��c(A)��c(Na+)����Һ����ĵ������������c(Na+)��c(H+)������Һ������Ũ�ȴ�С˳����c(A)��c(Na+)��c(H+)��c(OH)��

 ���100�ֵ�Ԫ�Ż�������ϵ�д�
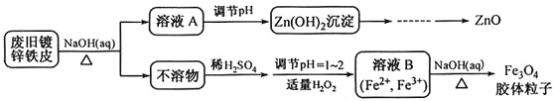
���100�ֵ�Ԫ�Ż�������ϵ�д�����Ŀ�����÷Ͼ�п��Ƥ�Ʊ�����Fe3O4�������Ӽ�������ZnO���Ʊ�����ͼ���£�
��֪��Zn���������������Al����������������ơ���ش��������⣺
��1����NaOH��Һ�����Ͼ�п��Ƥ��������_________________________________________��
A��ȥ������ | B���ܽ��п�� | C��ȥ������ | D���ۻ� |
��2��������ҺA��pH�ɲ���Zn(OH)2������Ϊ�Ƶ�ZnO����������������______________��
��3������ҺB�Ƶ�Fe3O4�������ӵĹ����У������ͨ��N2��ԭ����___________________��
��4��Fe3O4���������ܷ��ü�ѹ���˷�ʵ�ֹ�Һ���룿____________������������������������������_________________________________________________��
��5�����ظ���ط���һ��������ԭ�ζ������ɲⶨ����Fe3O4�еĶ�������������������Ũ��Ϊ0.01000 mol��L��1��K2Cr2O7����Һ250 mL��Ӧȷ��ȡ_______g K2Cr2O7(����4λ��Ч���֣���֪M(K2Cr2O7)��294.0 g��mol��1)��
���Ƹñ���Һʱ�����������в���Ҫ�õ�����_____________�����ñ�ű�ʾ����
�ٵ�����ƽ ���ձ� ����Ͳ �ܲ����� ������ƿ ��ͷ�ι� ����Һ��
��6���ζ������У�����ζ�ǰװ��K2Cr2O7����Һ�ĵζ��ܼ��첿�������ݣ����ζ�������������ʧ����ⶨ�����__________������ƫ��������ƫС����������������