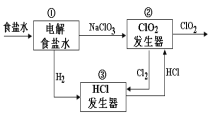
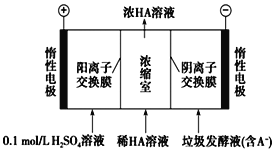
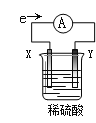
��Ŀ����
����Ŀ�������仯����������������Ӧ�ù㷺�������죨Fe2O3������Ϊ���ϣ����ӹ�ҵ����һ��Ũ�ȵ�FeCl3��Һ��ʴ����ͭ���ľ�Ե�壬�Ƴ�ӡˢ��·�塣 aFe2��SO4�� 3��b��NH4�� 2SO4��cH2O����������泥���������������ˮ����ҵѭ��ˮ�ľ���������
��1������һ����Fe2O3��Fe3O4�Ļ������Ʒ�����n��Fe��:n��O��=1:1.375,�����Ʒ��Fe2O3�����ʵ�������Ϊ___________�����������2λ��Ч���֣�
��2��CuO��Fe2O3�Ļ����9.6 g�ڸ�������������CO��ַ�Ӧ����Ӧ��ȫ��������100mL 1.2mol/L Ba��OH��2 ��Һ���գ�����15.76 g��ɫ�������������������Һ�е����ʵĻ�ѧʽΪ__________���������CuO��Fe2O3�����ʵ���֮��Ϊ___________��
��3����ȡij���������Ʒ7.00 g����������ˮ���Ƴ�100 mL��Һ���ֳ����ȷݣ�������һ���м�������NaOH��Һ������ϴ�ӵõ�1.07 g����������һ����Һ�м��뺬0.025 molBa ��NO3��2����Һ��ǡ����ȫ��Ӧ�����������淋Ļ�ѧʽ_________��
��4���ֽ�һ�����ͭ���ľ�Ե�����800mL 3mol/L��FeCl3��Һ�У�һ��ʱ�������·��ȡ��������Һ�м�������56.0 g����ַ�Ӧ��ʣ�����51.2 g����������Һ�����ʵ����ʵ���Ũ��_________�����Է�Ӧǰ����Һ����ı仯����
���𰸡�33% Ba(HCO3)2 1:1 Fe2��SO4��32��NH4��2SO42H2O FeCl2 4.25 mol/L��CuCl2 0.25 mol/L
��������
��1����Fe2O3��Fe3O4�Ļ��������ʵ����ֱ���x��y����n(Fe):n(O)=(2x+3y):(3x+4y)=1:1.375����x:y=1:2�������Ʒ��Fe2O3�����ʵ�������Ϊ![]() =33%���ʴ�Ϊ��33%��
=33%���ʴ�Ϊ��33%��
��2��100mL1.2mol/LBa(OH)2�����ʵ���Ϊ0.12mol������15.76g��0.08mol��ɫ����̼�ᱵ����������̼���㣬����̼Ԫ���غ㣬�����Ķ�����̼��0.08mol����ʱ���ݻ�����������㣬���ɵĶ�����̼�϶�����0.08mol���ʶ�����̼��������ʱ��Һ������ΪBa(HCO3)2����Ba(OH)2+ CO2 = BaCO3��+H2O��BaCO3+ CO2+H2O = Ba(HCO3)2�ã�����̼ԭ���غ��n(CO2)=0.08mol+(0.012mol-0.08mol)��2=0.16mol����������CuO��Fe2O3�����ʵ����ֱ�Ϊxmol��ymol����x+3y=0.16��80x+160y=9.6�����x=0.04mol��y=0.04mol�����Ի������CuO��Fe2O3�����ʵ���֮��Ϊ1:1���ʴ�Ϊ��Ba(HCO3)2��1:1��
��3����ȡ7.00g��Ʒ����������ˮ���ó�100mL��Һ,�ֳ����ȷݣ�������һ���м�������NaOH��Һ������ϴ�ӵõ�1.07g������ӦΪFe(OH)3��n(Fe(OH)3)=![]()
![]() =0.01mol������һ����Һ�м���0.025molBa(NO3)2��Һ��ǡ����ȫ��Ӧ����n(SO42)=0.025mol������7.00g��Ʒ�к���Fe2(SO4)30.01mol�����к���n(SO42)Ϊ0.05mol����(NH4)2SO4Ϊ0.05mol0.01��3mol=0.02mol����m(H2O)=7.00g0.01mol��400g/mol0.02mol��132g/mol=0.72g��n(H2O)=
=0.01mol������һ����Һ�м���0.025molBa(NO3)2��Һ��ǡ����ȫ��Ӧ����n(SO42)=0.025mol������7.00g��Ʒ�к���Fe2(SO4)30.01mol�����к���n(SO42)Ϊ0.05mol����(NH4)2SO4Ϊ0.05mol0.01��3mol=0.02mol����m(H2O)=7.00g0.01mol��400g/mol0.02mol��132g/mol=0.72g��n(H2O)=![]() =0.04mol��n(Fe2(SO4)3):n((NH4)2SO4):n(H2O)=0.01:0.02:0.02=1:2:2�����Ի�ѧʽΪFe2��SO4��32��NH4��2SO42H2O���ʴ�Ϊ��Fe2��SO4��32��NH4��2SO42H2O��
=0.04mol��n(Fe2(SO4)3):n((NH4)2SO4):n(H2O)=0.01:0.02:0.02=1:2:2�����Ի�ѧʽΪFe2��SO4��32��NH4��2SO42H2O���ʴ�Ϊ��Fe2��SO4��32��NH4��2SO42H2O��
��4���Ȼ����ܽ�ͭ�����Ȼ��������Ȼ�ͭ����Ӧ����Һ��Һ�м������ۣ�ʣ���������С��Fe�۵����������ܽ�Cu����Һ�л���ʣ���FeCl3�����������ȷ�����2Fe3++Fe=3Fe2+��Ȼ������Fe+Cu2+=Fe2++Cu�������й���ʣ�࣬��Fe3+��ȫ��Ӧ����Һ�к�������FeCl2�����跴Ӧ����Һ��û��CuCl2�������������غ��֪��Һ��FeCl2Ϊ![]() =3.6mol��������FeӦΪ3.6mol0.8L��3mol/L=1.2mol����ʵ�ʼ���FeΪ
=3.6mol��������FeӦΪ3.6mol0.8L��3mol/L=1.2mol����ʵ�ʼ���FeΪ![]() =1mol<1.2mol������Һ��һ������CuCl2������Feԭ���غ㣬��Һn(FeCl2)=n(FeCl3)+n(Fe)=0.8L��3mol/L+1mol=3.4mol������Һc(FeCl2)=
=1mol<1.2mol������Һ��һ������CuCl2������Feԭ���غ㣬��Һn(FeCl2)=n(FeCl3)+n(Fe)=0.8L��3mol/L+1mol=3.4mol������Һc(FeCl2)=![]() =4.25mol/L�������������غ㣬n(CuCl2)=
=4.25mol/L�������������غ㣬n(CuCl2)=![]() ��(0.8L��3mol/L��33.4mol��2)=0.2mol������Һc(CuCl2)=
��(0.8L��3mol/L��33.4mol��2)=0.2mol������Һc(CuCl2)=![]() =0.25mol/L���ʴ�Ϊ��FeCl2 4.25 mol/L��CuCl2 0.25 mol/L��
=0.25mol/L���ʴ�Ϊ��FeCl2 4.25 mol/L��CuCl2 0.25 mol/L��

 ѧ���������ν��Ͼ���ѧ������ϵ�д�
ѧ���������ν��Ͼ���ѧ������ϵ�д� Happy holiday���ּ��������ҵ�㶫���������ϵ�д�
Happy holiday���ּ��������ҵ�㶫���������ϵ�д�