��Ŀ����
7��ijͬѧ����ʵ���о�ʱ��������1.0mol•L-1Ba��OH��2��Һ����ֻ�ҵ��ڿ����б�¶�Ѿõ�Ba��OH��2•8H2O�Լ�����ѧʽ����315������������������Һʱ������ȡ�Լ���ˮ�н������ܽ⣬�ձ��д��ڴ���δ���Ϊ̽����ԭ��ͬѧ���Ba��OH��2•8H2O��283K��293K��303Kʱ���ܽ�ȣ�g/100g H2O���ֱ�Ϊ2.5��3.9��5.6����1���ձ���δ������ܽ�ΪBaCO3��������Ba��OH��2•8H2O��CO2����ת��ΪBaCO3��
��2�������Լ��ɴ���Ba��OH��2•8H2O������BaCO3��ɣ����ʵ�鷽�������гɷּ�
�飮д��ʵ�鲽�衢Ԥ������ͽ��ۣ��������ǽᾧˮ�ļ��飻����ʱBaCO3������Һ��pH=9.6��
��ѡ�Լ���������ϡ���ᡢϡ���ᡢNaOH��Һ������ʯ��ˮ��pH�ơ��ձ����Թܴ��������ܡ��ι�
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ�����Լ��ڽྻ�ձ��У�������������ˮ����ֽ��裬���ã����ˣ�����Һ�ͳ����� | |
| ����2��ȡ������Һ���Թ��У��μ�ϡ���ᣮ | |
| ����3��ȡ��������1�еij������Թ��У�ȡ��������1�еij������Թ��У��μ�ϡ���ᣬ�ô��������������Թܣ��ѵ����ܲ���װ�г���ʯ��ˮ���ձ��У� | |
| ����4��ȡ������Һ���ձ��У���pH�Ʋ��� pHֵ |
������250mLԼ0.1mol•L-1 Ba��OH��2��Һ��ȷ��ȡw�������������ձ��У�����������ˮ���ܽ⣬����Һת��250ml������ƿ�У�ϴ�ӣ����ݣ�ҡ�ȣ�
�ڵζ���ȷ��ȡ25.00mL����Ba��OH��2��Һ����ƿ�У��μ�ָʾ������0.1980���0.0210������0.0536������0.1980����1.5000����mol•L-1����װ��50mL��ʽ�ζ��ܣ��ζ����յ㣬��¼���ݣ��ظ��ζ�2�Σ�ƽ����������V mL��
�ۼ���Ba��OH��2•8H2O����������=$\frac{0.25L��\frac{0.198V}{50}mol��315g/mol}{wg}$��100%��ֻ�г���ʽ���������㣩��
��4�������£����ܣ���ܡ����ܡ��� ����1.0 mol•L-1 Ba��OH��2��Һ��
���� ��1�����������ܹ��������̼��Ӧ����̼�ᱵ��
��2������3��ȡ��������1�еij������Թ��У��μ�ϡ���ᣬ�ô��������������Թܣ��ѵ����ܲ���װ�г���ʯ��ˮ���ձ��У�BaCO3�ܺ����ᷴӦ���ɵĶ�����̼������ʹʯ��ˮ����ǣ���Ԥ������ͽ��ۣ��Թ������������ɣ��ձ��еij���ʯ��ˮ����ǣ���ϲ���2˵��������BaCO3��
����4��ȡ������Һ���ձ��У���pH�Ʋ���pHֵ��Ԥ������ͽ��ۣ�pH��9.6��˵���д�����OH-���ۺ����沽���֪�Լ��ɴ���Ba��OH��2•8H2O������BaCO3��ɣ����������
��3������������һ�����ʵ���Ũ����Һ�����Ʋ���ѡ����ʵ��������ݴ˽��
������������ܶ���50ml������50mL�����������СŨ�ȣ�Ӧ����ѡ������СŨ�ȶ��ҽӽ��ģ�
����250mL��Һ������������Ũ��Ϊcmol/L���������ĵ������з��̼���c��ֵ����������250mL��Һ���������������ʵ������ݴ˼�����Ʒ��Ba��OH��2•8H2O������������
��4�����������������ܽ�ȹ��㱥����Һ�����ʵ���Ũ�ȣ��ݴ��жϣ�
��� �⣺��1���ձ���δ�����ΪBaCO3��������Ba��OH��2•8H2O��CO2����ת��ΪBaCO3��
�ʴ�Ϊ��Ba��OH��2•8H2O��CO2����ת��ΪBaCO3��
��2������3��ȡ��������1�еij������Թ��У��μ�ϡ���ᣬ�ô��������������Թܣ��ѵ����ܲ���װ�г���ʯ��ˮ���ձ��У�BaCO3�ܺ����ᷴӦ���ɵĶ�����̼������ʹʯ��ˮ����ǣ���Ԥ������ͽ��ۣ��Թ������������ɣ��ձ��еij���ʯ��ˮ����ǣ���ϲ���2˵��������BaCO3��
����4��ȡ������Һ���ձ��У���pH�Ʋ���pHֵ��Ԥ������ͽ��ۣ�pH��9.6��˵���д�����OH-���ۺ����沽���֪�Լ��ɴ���Ba��OH��2•8H2O������BaCO3��ɣ����������
�ʴ�Ϊ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ�����Լ��ڽྻ�ձ��У�������������ˮ����ֽ��裬���ã����ˣ�����Һ�ͳ����� | ---------- |
| ����2��ȡ������Һ���Թ��У��μ�ϡ���ᣮ | �а�ɫ�������ɣ�˵����Ba2+ |
| ����3��ȡ��������1�еij������Թ��У��μ�ϡ���ᣬ�ô��������������Թܣ��ѵ����ܲ���װ�г���ʯ��ˮ���ձ��� | �Թ������������ɣ��ձ��еij���ʯ��ˮ����ǣ���ϲ���2˵��������BaCO3 |
| ����4��ȡ������Һ���ձ��У���pH�Ʋ��� pHֵ | pH��9.6��˵���� ������OH-���ۺ����沽���֪�Լ��ɴ���Ba��OH��2•8H2O������BaCO3��ɣ�������� |
�ʴ�Ϊ���ܽ⣻250ml������ƿ��
������������ܶ���50ml������50mL�����������СŨ�ȣ����������СŨ��Ϊamol/L����
0.025L��0.1mol/L��2=amol/L��0.05L�����a=0.1mol/L��Ӧ����ѡ������СŨ�ȶ��ҽӽ��ģ���ѡ��0.1980mol/L�����ᣬ
�ʴ�Ϊ��0.1980��
����250mL��Һ������������Ũ��Ϊcmol/L����
0.025L��cmol/L��2=0.1980mol/L��V��10-3L�����c=$\frac{0.198V}{50}$��
��Ba��OH��2•8H2O����������=$\frac{0.25L��\frac{0.198V}{50}mol��315g/mol}{wg}$��100%��
�ʴ�Ϊ��$\frac{0.25L��\frac{0.198V}{50}mol��315g/mol}{wg}$��100%��
��4�������£������������ܽ��Ϊ3.9g����Һ�����ʵ���Ũ��Լ��$\frac{\frac{3.9g}{171g/mol}}{0.1L}$=0.23mol/L�����������²�������1.0mol/L��1Ba��OH��2��Һ���ʴ�Ϊ�����ܣ�
���� ���⿼�����ʳɷ��뺬���ⶨ����ʵ�鷽������������ơ���Һ���ơ��к͵ζ�����ѧ����ȣ���Ŀ�ۺ���ǿ���ѶȽϴ��ض�ʵ�鷽������뻯ѧ���㣮

Na2O2+CO2��g���TNa2CO3��s��+$\frac{1}{2}$O2��g����H=-226kJ/mol
���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ���ǣ�������
| A�� | Na2O2��s����CO2��g����Ӧʱ���ų�452kJ����ʱ��ת�Ƶ�����Ϊ1.204��l023 | |
| B�� | CO��ȼ����Ϊ��H=-566kJ/mol | |
| C�� | CO��g����Na2O2 ��S����Ӧ���Ȼ�ѧ����ʽΪCO��g��+Na2O2��s���TNa2CO3��s����H=-509kJ/mol | |
| D�� | 2Na2O2��s��+2CO2��s���T2Na2CO3��s��+O2��g����H=-452kJ/mol |
| A�� | ����ȼ�� | B�� | ����Һ�� | C�� | ̼��Ʒֽ� | D�� | Ũ����ϡ�� |
| A�� |  ����ʳ��ˮ | B�� |  ����Ͳ��ȡҺ�� | C�� |  ����Һ��pH | D�� |  ����NH3 |
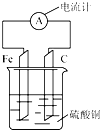 ����Ƭ��̼������ͼ��ʾ��ʽ��������ͭ��Һ�У�������ָ�뷢��ƫת��������Ը�װ�õ�˵����ȷ���ǣ�������
����Ƭ��̼������ͼ��ʾ��ʽ��������ͭ��Һ�У�������ָ�뷢��ƫת��������Ը�װ�õ�˵����ȷ���ǣ�������| A�� | ��Ƭ������ | |
| B�� | һ��ʱ���̼���������� | |
| C�� | ��װ���ܽ�����ת��Ϊ��ѧ�� | |
| D�� | ���·�е�����̼����������������������Ƭ |
| A�� | FeO | B�� | Fe2O3 | C�� | FeCl3 | D�� | Fe��OH��3 |
| ��Է������� | �ܶ�/��g•cm-3�� | �е�/�� | ˮ���ܽ��� | |
| ������ | 88 | 0.8123 | 131 | �� |
| ���� | 60 | 1.0492 | 118 | �� |
| ���������� | 130 | 0.8670 | 142 | ���� |
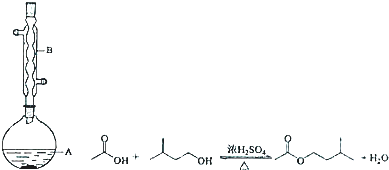
����A�м���4.4g���촼��6.0g���ᡢ����Ũ���
�ڻ�������A������50min��
�۷�ӦҺ�������º����������У��ֱ�������ˮ������̼��������Һ��ˮϴ�ӣ�
�ֳܷ��IJ������������ˮMgSO4���壬����Ƭ�̣����˳�ȥMgSO4���壻
�ݽ����������ռ�140��143����֣�������������2.6g��
��ش��������⣺
��1����������Ӧ��ԭ����������$\frac{23}{26}$���÷�����ʾ����
��2��ʵ��������B������������������������е��������Ƿ�Һ©����
��3����ͬѧ��ʼ����A���ִ�ǰ��������һ�����ص���©����β��ȣ�װ����ȴ���ٲ�����Ƭ��ʯ�����Ƭ��
��4��������м�����ˮMgSO4��Ϊ�˳�ȥij�����ʣ���μ���������Ƿ����������ʵ�鷽����ȡ�����ֲ���������м���������ˮ����ͭ����������������ˮ�ѳ�����
��5��������У����¶ȼ�ˮ�����λ�ù��ߣ������ռ���ֵķе���ڣ�����ڡ����ڡ���Ŀ�����ķе㣮
��6����ʵ��IJ�����d�����ţ��� a��30% ��b��40% c��50% d��60%