��Ŀ����
����Ŀ����������(H2O2)��ҽҩ�����������õȷ����й㷺��Ӧ�á��ش��������⣺
��1�������Ʊ�H2O2ʹ�õ�ԭ����������ˮ��BaO2��ϡ���ᣬ������Ӧ�Ļ�ѧ����ʽ��_____��BaO2�ĵ���ʽΪ��__________��
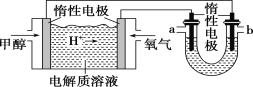
��2��Ŀǰ��ҵ�Ʊ�H2O2����Ҫ����������������Ӧ�������£�
![]()
��֪���һ������Ƿ�Ӧ�еĴ�����
������������ԭ�ϵ����ʵ���֮��Ϊ_____________��
�ڲ���a��__________________��
����������Һ�ijɷ���______________________��
��3��������������Ũ�ȳ��õķ����ǵζ�����ij�βⶨʱ��ȡ20. 00mL����������Ʒ��������������ữ����0. l000mol/L��KMnO4����Һ�ζ����յ㣬����10. 00mL���ζ�ʱ������Ӧ�����ӷ���ʽΪ____������Ʒ��H2O2�����ʵ���Ũ��Ϊ_______��
���𰸡�BaO2+H2SO4=BaSO4��+H2O2 ![]() 1:1 ��ȡ��Һ �л��ܼ��һ����� 2MnO4��+5H2O2+6H+=2Mn2++5O2��+8H2O 0.1250mol/L
1:1 ��ȡ��Һ �л��ܼ��һ����� 2MnO4��+5H2O2+6H+=2Mn2++5O2��+8H2O 0.1250mol/L
��������
��1��������ˮ��BaO2��ϡ���ᷴӦ����H2O2��BaSO4��������������Ϊ���ӻ����
��2��������ͼ��֪���һ����������л��ܼ��õ��һ��������л���Һ���������һ��������л���Һ�ڴ��������·�Ӧ�����⻯Һ�һ����������һ�����������������Ӧ���ɺ����һ��������л���Һ��˫��ˮ������Һ��������Һ�м�������ˮ����ȡ��Һ�õ�ϡ˫��ˮ�ͺ����һ��������л���Һ�Ļ���Һ��
��3��˫��ˮ��������������KMnO4����������ԭ��Ӧ���������̡�������ˮ�����ݷ�Ӧ����ʽ�ɼ�����Ʒ��H2O2�����ʵ���Ũ�ȡ�
��1��������ˮ��BaO2��ϡ���ᷴӦ����H2O2��BaSO4��������Ӧ�Ļ�ѧ����ʽΪBaO2+H2SO4=BaSO4��+H2O2����������Ϊ���ӻ���������������������ͨ�����Ӽ���ϣ���ԭ��֮��ͨ�����ۼ���ϣ����������ĵ���ʽ��![]() ���ʴ�Ϊ��BaO2+H2SO4=BaSO4��+H2O2��
���ʴ�Ϊ��BaO2+H2SO4=BaSO4��+H2O2��![]() ��
��
��2�������ݷ�Ӧԭ����֪���������Ʊ�˫��ˮ���������ĵ�ԭ��������������������˫��ˮ�Ļ�ѧʽ��֪��ԭ�ϵ����ʵ���֮��Ϊ1��1���ʴ�Ϊ��1:1��
����������ˮ����в���a�õ�ϡ˫��ˮ����������Һ����������ԭ����������ҺӦ��Ϊ�һ��������ʲ����ǽ��һ�������ˮ�з��������Ӧ������ȡ��Һ���ʴ�Ϊ����ȡ��Һ��
����������Һ�ijɷ����л��ܼ����һ��������ʴ�Ϊ���л��ܼ����һ�������
��3��˫��ˮ��������������KMnO4����������ԭ��Ӧ���������̡�������ˮ����Ӧ�����ӷ���ʽΪ2MnO4��+5 H2O2+6H+=2Mn2++5O2��+8H2O�����ݷ�Ӧ����ʽ�ɵ���Ʒ��H2O2�����ʵ���Ũ��Ϊ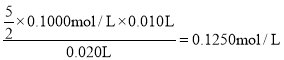 ���ʴ�Ϊ��2MnO4��+5 H2O2+6H+=2Mn2++5O2��+8H2O��0.1250mol/L��
���ʴ�Ϊ��2MnO4��+5 H2O2+6H+=2Mn2++5O2��+8H2O��0.1250mol/L��

 Ӧ������ҵ��ϵ�д�
Ӧ������ҵ��ϵ�д�����Ŀ��������X������Y�������Z��ˮ��Һ�����һ��ʱ����ټ���W����ʹ��Һ�ָ������ǰ��״̬�����������һ����(����)
ѡ�� | ����X | ����Y | Z | W |
A | C | Fe | KCl | KOH |
B | Pt | Cu | CuSO4 | Cu��OH)2��Һ |
C | C | C | H2SO4 | H2O |
D | Ag | Fe | AgNO3 | AgNO3���� |
A. AB. BC. CD. D