��Ŀ����
����Ŀ������˵������ȷ���ǣ� ��
A.25��ʱij��Һ��ˮ�������c(H+)��1.0��1012 mol��L1����pHһ����12
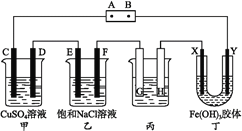
B.ij�¶��£���ˮ��ͨ��CO2������CO2��ͨ�룬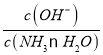 ��������
��������
C.���º����£���ӦX(g)��3Y(g) ![]() 2Z(g)����ʼ����3 mol X��3 mol Y����X�������������ʱ����Ӧ�ﵽƽ��
2Z(g)����ʼ����3 mol X��3 mol Y����X�������������ʱ����Ӧ�ﵽƽ��
D.ij�¶��£���pH��6������ˮ�м���NaHSO4���壬�����¶Ȳ��䣬�����Һ��pHΪ2�����¶��¼�������pH��10��NaOH��Һ��ʹ��Ӧ�����Һǡ�ó�����
���𰸡�D
��������
A. ��Ȼ��ˮ������c��H+��=1.0��10-12mol/L������ˮ������c��OH-��==1.0��10-12mol/L����c��H+����c��OH-��=1.0��10-14mol/L������ԭ������Һ��c��H+��=1.0��10-2mol/L����c��OH-��=1.0��10-2mol/L������Һ���ܳ���ǿ���ǿ���ԣ�ǿ��ʱpH=2��ǿ��ʱpH=12����A����
B. NH3��H2O ![]() NH4����OH-��ͨ��CO2ƽ�������ƶ���c(NH4+)��������ƽ�ⳣ�����䣬��
NH4����OH-��ͨ��CO2ƽ�������ƶ���c(NH4+)��������ƽ�ⳣ�����䣬��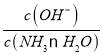 =
=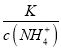 ���ϼ�С����B����
���ϼ�С����B����
C. ���º����£���ӦX(g)��3Y(g) ![]() 2Z(g)����ʼ����3 mol X��3 mol Y��������ʽ�����֪��X���������һֱ��50%����C����
2Z(g)����ʼ����3 mol X��3 mol Y��������ʽ�����֪��X���������һֱ��50%����C����
D. ����ˮ��pH��6������Kw=1��10-12��Ӧ��������Ũ��Ϊ0.01mol L-1������������Һ����Ӧ��������pH=10��NaOH��Һ����ʹ����Һǡ�ó����ԣ���D��ȷ��
��ȷ����D��

 ��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
��ĩ���䵥Ԫ�����ิϰ��ϵ�д�