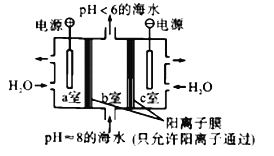
��Ŀ����
����Ŀ�����÷ϱ���(��Ҫ�ɷ�ΪBaS2O3��������SiO2)Ϊԭ�������ߴ�������������������

��֪:Kap(BaS2O3)=6.96��10-11��Kap(BaF2)=1.0��10-6
(1)����ٳ�����SO2�������е���ɫ�������ɣ��÷�Ӧ�����ӷ���ʽΪ______________��
(2)����ڵ�Ŀ�����к���������������NaOH��Һ���˹�������ԭ����__________(�����ӷ���ʽ��ʾ)��
(3)��Һ����Ҫ�ɷ���BaCl2������������NaCl���ܽ���������±���
�¶� | 20�� | 40�� | 60�� | 80�� | 100�� |
NaCl | 36.0g | 36.6g | 37.3g | 39.0g | 39.8g |
BaCl2 | 35.8g | 40.8g | 46.2g | 52.5g | 59.4g |
������˲���_____ (���������ᾧ���������½ᾧ��)��
(4)��ҵ�Ͽ��ð�ˮ����SO2����ͨ�����ʹ��ת��Ϊ�̬���ʡ���ת�����������뻹ԭ�������ʵ���֮��Ϊ__________��
(5)���������BaF2�ķ�Ӧ����Ϊ____________��
�����÷�Ӧ�¶ȹ��ߣ��������c(F-)���͵�ԭ����__________��
���о��������ʵ�����NH4F�ı������������BaF2�IJ��ʺʹ��ȣ���Ũ��Ϊ0. 1mol��L-1��BaCl2��Һ��0.22 mol��L-1NH4F��Һ����������������Һ��c(Ba2+)=__________ mol��L-1��
���𰸡� BaS2O3+2H+=Ba2++S��+SO2��+H2O 2OH-+SiO2=SiO32-+H2O ���½ᾧ 1:2 ���ֽⷴӦ �¶Ƚϸߴٽ�F-ˮ��,ʹc(F-)���� 0.01
��������������(1)��������ͼ��������зϱ����е�BaS2O3�����ᷴӦ����SO2������
(2)����ڵ�Ŀ�����к��������ᣬ�������������ƻ���������跴Ӧ��
(3)����BaCl2��NaCl�ܽ�����ݿ�֪���Ȼ������ܽ�����¶ȵı仯���Ȼ��Ƶ��ܽ�����¶ȵı仯��ͬ��
(4)ת����������Ϊ��������ԭ��ΪSO32-��SO2�����ݵ�ʧ�����غ������
(5)���������BaF2�Ļ�ѧ��Ӧ����ʽΪBaCl2+2NH4F=BaF2+2NH4Cl���������ˮ�����¶ȵ�Ӱ�죬�¶�Խ�ߣ�ˮ��̶�Խ�����ڸ���BaCl2+2NH4F=BaF2+2NH4Cl���㷴Ӧ����Һ��c(F-)���ٸ���Kap(BaF2)����c(Ba2+)��
��⣺(1)������зϱ���(��Ҫ�ɷ�ΪBaS2O3��������SiO2)�е�BaS2O3�����ᷴӦ������SO2�⣬���е���ɫ���������ɣ���Ӧ�����ӷ���ʽΪBaS2O3+2H+=Ba2++S��+SO2��+H2O���ʴ�Ϊ��BaS2O3+2H+=Ba2++S��+SO2��+H2O��
(2)����ڵ�Ŀ�����к��������ᣬ����NaOH��Һ���˹������������������ƻ���������跴Ӧ�����ӷ���ʽΪ2OH-+SiO2=SiO32-+H2O���ʴ�Ϊ��2OH-+SiO2=SiO32-+H2O��
(3)��Һ����Ҫ�ɷ���BaCl2������������NaCl�����ݶ����ܽ�����ݿ�֪���Ȼ������ܽ�����¶ȵı仯�Ƚ����ԣ����Ȼ��Ƶ��ܽ�����¶ȵı仯��������˲��ý��½ᾧ���ʴ�Ϊ�����½ᾧ��
(4)��ҵ�Ͽ��ð�ˮ����SO2����ͨ�����ʹ��ת��Ϊ�̬���ʣ���ת����������Ϊ��������ԭ��ΪSO32-��SO2�����ݵ�ʧ�����غ㣬�������ʵ���֮��Ϊ1:2���ʴ�Ϊ��1:2��
(5)���������BaF2�Ļ�ѧ��Ӧ����ʽΪBaCl2+2NH4F=BaF2+2NH4Cl�����ڸ��ֽⷴӦ���ʴ�Ϊ�����ֽⷴӦ��
�����÷�Ӧ�¶ȹ��ߣ��¶Ƚϸߴٽ�F-ˮ�⣬ʹc(F-)���ͣ��ʴ�Ϊ���¶Ƚϸߴٽ�F-ˮ�⣬ʹc(F-)������
��������Һ��c(F-)=![]() ��(0.22 mol��L-1-0.2 mol��L-1)=0.01 mol��L-1��c(Ba2+)=
��(0.22 mol��L-1-0.2 mol��L-1)=0.01 mol��L-1��c(Ba2+)=![]() =
=![]() = mol��L-1���ʴ�Ϊ��0.01��
= mol��L-1���ʴ�Ϊ��0.01��

 ��У����ϵ�д�
��У����ϵ�д�