ΧβΡΩΡΎ»ί
ΓΨΧβΡΩΓΩœ¬±μΈΣ‘ΣΥΊ÷ήΤΎ±μΒΡ“Μ≤ΩΖ÷Θ§”Ο«κΜ·―ß”Ο”οΜΊ¥π”–ΙΊΈ ΧβΘΚ
IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 | |
2 | E | F | ||||||
3 | A | C | D | G | R | |||
4 | B | H |
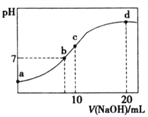
(1)CΚΆHΒΡ‘ΣΥΊΖϊΚ≈ «______ΚΆ_______ΘΜ
(2)±μ÷–A”κFΒΡ«βΜ·ΈοΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ___________Θ§EΒΡΒΞ÷ ”κΤδœ¬“Μ÷ήΤΎΆ§Ήε‘ΣΥΊΒΡΉνΗΏΦέ―θΜ·Έο‘ΎΗΏΈ¬œ¬Ζ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ___________ΘΜΉν≤ΜΜνΤΟΒΡ‘ΣΥΊ________(Χν–¥‘ΣΥΊΖϊΚ≈)
(3)±μ÷–ΥυΝ–‘ΣΥΊ÷–ΉνΗΏΦέ―θΜ·ΈοΕ‘”ΠΥ°Μ·ΈοΦν–‘Ήν«ΩΒΡ «___________Θ§Υα–‘Ήν«ΩΒΡ «______ (Χν–¥Μ·―ß Ϋ)
(4)±μ÷–Ρή–Έ≥…ΝΫ–‘«β―θΜ·ΈοΒΡ‘ΣΥΊ «__________(Χν–¥‘ΣΥΊΖϊΚ≈)Θ§–¥≥ωΗΟ‘ΣΥΊΒΡ«β―θΜ·Έο”κAΒΡΉνΗΏΦέ―θΜ·ΈοΕ‘”ΠΥ°Μ·ΈοΖ¥”ΠΒΡάκΉ”ΖΫ≥Χ Ϋ__________________ΓΘ
ΓΨ¥πΑΗΓΩMg Br 2Na+2H2O=2NaOH+H2Γϋ 2C+SiO2 ![]() Si+2COΓϋ Ar KOH HClO4 Al Al(OH)3+OH-=AlO
Si+2COΓϋ Ar KOH HClO4 Al Al(OH)3+OH-=AlO![]() +2H2O
+2H2O
ΓΨΫβΈωΓΩ
ΗυΨίΗς‘ΣΥΊ‘Ύ÷ήΤΎ±μ÷–ΒΡΈΜ÷ΟΩ…÷ΣAΘΚNaΘ§BΘΚKΘ§CΘΚMgΘ§DΘΚAlΘ§EΘΚCΘ§FΘΚOΘ§GΘΚClΘ§HΘΚBrΘ§RΘΚArΓΘ
(1)CΚΆHΒΡ‘ΣΥΊΖϊΚ≈ «MgΓΔBrΘΜ
(2)AΈΣNaΘ§FΒΡ«βΜ·ΈοΈΣH2OΘ§Μ·―ßΖΫ≥Χ ΫΈΣ2Na+2H2O=2NaOH+H2ΓϋΘΜEΒΡΒΞ÷ ΈΣCΘ§œ¬“Μ÷ήΤΎΆ§Ήε‘ΣΥΊΈΣSiΘ§ΉνΗΏΦέ―θΜ·ΈοΈΣSiO2Θ§Μ·―ßΖΫ≥Χ ΫΈΣ2C+SiO2 ![]() Si+2COΓϋΘΜΉν≤ΜΜνΤΟΒΡ‘ΣΥΊΈΣArΘΜ
Si+2COΓϋΘΜΉν≤ΜΜνΤΟΒΡ‘ΣΥΊΈΣArΘΜ
(3)ΉνΗΏΦέ―θΜ·ΈοΕ‘”ΠΥ°Μ·ΈοΦν–‘«Ω»θ «Ε‘”ΎΫπ τ–‘ΒΡΩΦ≤ιΘ§Ϋπ τ–‘‘Ϋ«ΩΘ§Φν–‘‘Ϋ«ΩΘ§Ήσœ¬Ϋ«‘ΣΥΊΫπ τ–‘Ήν«ΩΘ§Ι Ήν«ΩΒΡ «KOHΘ§ΕχΥα–‘ «Ε‘Ζ«Ϋπ τ–‘ΒΡΩΦ≤ιΘ§Ζ«Ϋπ τ–‘‘Ϋ«ΩΘ§Υα–‘‘Ϋ«ΩΘ§”“…œΫ«‘ΣΥΊΖ«Ϋπ τ–‘Ήν«ΩΘ§OΆ®≥Θ≤Μœ‘’ΐΦέΘ§Ι Ήν«ΩΒΡ «HClO4ΘΜ
(4)Ρή–Έ≥…ΝΫ–‘«β―θΜ·ΈοΒΡ‘ΣΥΊ «AlΘ§Τδ«β―θΜ·Έο «Al(OH)3Θ§AΒΡΉνΗΏΦέ―θΜ·ΈοΕ‘”ΠΥ°Μ·Έο «NaOHΘ§Ζ¥”ΠάκΉ”ΖΫ≥Χ ΫΈΣAl(OH)3+OH-=AlO![]() +2H2OΓΘ
+2H2OΓΘ

 ΚηΆΦΆΦ ιΚ°ΦΌΉς“ΒΦΌΤΎΉς“ΒΦΣΝ÷¥σ―ß≥ωΑφ…γœΒΝ–¥πΑΗ
ΚηΆΦΆΦ ιΚ°ΦΌΉς“ΒΦΌΤΎΉς“ΒΦΣΝ÷¥σ―ß≥ωΑφ…γœΒΝ–¥πΑΗ