��Ŀ����
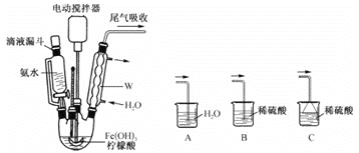
����Ŀ��ijʵ��С�����ü����ƣ�HCOONa���Ʊ�Na2S2O4���ⶨ��Ʒ�Ĵ��ȣ�ʵ��װ�ã��г֡����������ԣ���ͼ
�ش���������
��1��ʢ��Ũ�������������Ϊ________��д��ŨH2SO4��Cu��Ӧ�Ļ�ѧ����ʽ ________ ��
��2��SO2��HCOONa��NaOH��Ӧ����Na2S2O4��CO2�� �����ӷ���ʽΪ________��
��3��ͼ�����߿���װ�����ڼ����ݳ������е�CO2 �� a��b��c��ʢ�ŵ��Լ�����Ϊ________��________��________��
��4������ӦҺ�����ᴿ�õ�Na2S2O4��Ʒ��ȡ��Ʒmg����ˮ��������Ϊ100mL��ȡ25��00mL������ƿ�У�����NaOH��Һ��ָʾ������cmol��mol-1��K3[Fe(CN)6]����Һ���еζ�{4K3[Fe(CN)6]+2Na2S2O4+8NaOH=3K4[Fe(CN)6]+4Na2SO3+Na4[Fe(CN)6]+4H2O}���ζ����յ�ʱ�����ı�ҺVmL�����Ʒ�Ĵ���Ϊ________��д������ʽ����
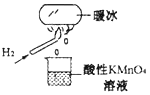
���𰸡���Һ©�� Cu+2H2SO4��Ũ�� ![]() CuSO4+SO2��+2H2O HCOO-+OH-+2SO2=S2O42-+CO2+H2O KMnO4��Һ�������������𰸣� Ʒ����Һ ����ʯ��ˮ
CuSO4+SO2��+2H2O HCOO-+OH-+2SO2=S2O42-+CO2+H2O KMnO4��Һ�������������𰸣� Ʒ����Һ ����ʯ��ˮ ![]() ��100%
��100%
��������
(1)����ͼʾ����ȷ�������ƣ�Ũ������������ԣ����������£� �ܽ�Cu������CuSO4 �� ������ԭΪSO2 �� �ݴ�д����Ӧ�Ļ�ѧ����ʽ��
(2)���������Ϣȷ����Ӧ����������ϵ���غ��ԭ���غ���д��Ӧ�����ӷ���ʽ��
(3)����CO2�ó���ʯ��ˮ�����������л��е�SO2��ʹ����ʯ��ˮ����ǣ�������ų�SO2�ĸ��ţ����SO2������ȷ��a��b���Լ���
(4)�������ı�Һ���������Ϸ�Ӧ�Ļ�ѧ����ʽ����mg��Ʒ��Na2S2O4�������Ӷ������Ʒ�Ĵ��ȡ�
(1)ͼ��ʢװŨ���������Ϊ��Һ©����Ũ������������ԣ����������£� �ܽ�Cu������CuSO4��������ԭΪSO2���÷�Ӧ�Ļ�ѧ����ʽΪ��Cu+2H2SO4(Ũ) ![]() CuSO4+SO2��+2H2O��
CuSO4+SO2��+2H2O��
(2)HCOONa��NaOH��Na2S2O4���ǿ�����ˮ����Һ�������ӷ���ʽ�пɲ𣬷�Ӧ�����У�������Na2S2O4�����CO2��H2O����ϵ���غ㡢ԭ���غ�ɵø÷�Ӧ�����ӷ���ʽΪ��HCOO����OH����2SO2=S2O42����CO2��H2O��
(3)����CO2�ó���ʯ��ˮ�����������л��е�SO2��ʹ����ʯ��ˮ����ǣ�������ų�SO2�ĸ��ţ�SO2���л�ԭ�ԣ�������ˮ������KMnO4��Һ���գ����a���Լ�Ϊ��ˮ������KMnO4��Һ��Ϊ����SO2�Ƿ���ȫ����������Ʒ����Һ�Ƿ���ɫ���м��飬���b���Լ�ΪƷ����Һ��������ó���ʯ��ˮ����CO2�� ���c���Լ�Ϊ����ʯ��ˮ��
(4)�ɷ�Ӧ�Ļ�ѧ����ʽ�ɵù�ϵʽ��2K3[Fe(CN)6]~Na2S2O4�������25.00mL��Ʒ��Һ������![]() �� ���mg��Ʒ������
�� ���mg��Ʒ������![]() �� �����ò�Ʒ�Ĵ���Ϊ
�� �����ò�Ʒ�Ĵ���Ϊ![]() ��
��