��Ŀ����
3��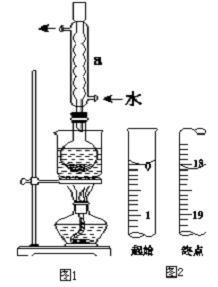 Na2S2O3����Ҫ�Ļ���ԭ�ϣ�������ˮ�������Ի���Ի������ȶ���
Na2S2O3����Ҫ�Ļ���ԭ�ϣ�������ˮ�������Ի���Ի������ȶ������Ʊ�Na2S2O3•5H2O
��Ӧԭ����Na2SO3��aq��+S��s��$\frac{\underline{\;\;��\;\;}}{\;}$Na2S2O3��aq��
ʵ�鲽�裺
�ٳ�ȡ20g Na2SO3����Բ����ƿ�У��ټ���80mL����ˮ����ȡ4g��ϸ����ۣ���3mL�Ҵ���ʪ������������Һ�У�
�ڰ�װʵ��װ�ã���ͼ1��ʾ�����ּг�װ����ȥ����ˮԡ���ȣ���60min��
�۳��ȹ��ˣ�����Һˮԡ����Ũ������ȴ����Na2S2O3•5H2O�������ˡ�ϴ�ӡ�����õ���Ʒ��
�ش����⣺
��1������ڷ�Ӧǰ���Ҵ���ʪ��Ŀ����ʹ������ڷ�ɢ����Һ�У�
��2������a�������������ܣ�������������������
��3����Ʒ�г�����δ��Ӧ��Na2SO3�⣬����ܴ��ڵ���������Na2SO4�������Ƿ���ڸ����ʵķ�����ȡ������Ʒ���ڹ���ϡ���ᣬ���ˣ�����Һ�м�BaCl2��Һ�����а�ɫ���������Ʒ�к���Na2SO4��
��4����ʵ��һ������ڼ��Ի����½��У������Ʒ���ƣ������ӷ�Ӧ����ʽ��ʾ��ԭ��S2O32?+2H+=S��+SO2��+H2O��
�ⶨ����
�����ò�Ʒ���500ml��Һ��ȡ����Һ20ml���Ե�����ָʾ������0.100 0mol•L-1��ı���Һ�ζ�����Ӧԭ��Ϊ2S2O32-+I2�TS4O62-+2I-
��5���ζ����յ�ʱ����Һ��ɫ�ı仯������ɫ��Ϊ��ɫ��
��6���ζ���ʼ���յ��Һ��λ����ͼ2�������ĵ�ı���Һ���Ϊ18.10mL�����ʵ��IJ���Ϊ72.4%����������λ��Ч���ݣ�
��Na2S2O3��Ӧ��
��7��Na2S2O3��ԭ�Խ�ǿ������Һ���ױ�Cl2������SO42-�����������ȼ����÷�Ӧ�����ӷ���ʽΪS2O32?+4Cl2+5H2O=2SO42?+8Cl?+10H+��
���� ��1�����������ˮ�������Ҵ����Ҵ�ʪ�����ʹ������ڷ�ɢ����Һ�У�
��2�����������ṹ����д�������ƣ����������������������ã�
��3������Na2S2O3��Na2SO3���л�ԭ�ԣ��ױ���������Na2SO4��������Һ�Ƿ���������ӽ����жϣ�
��4��S2O32-������������Һ���ܹ�����������ԭ��Ӧ�����������������
��5���ζ�ǰ��ҺΪ��ɫ���ζ������ⵥ��ʹ���۱�����
��6������ͼʾ�ĵζ�����Һ��������������ն�����Ȼ���������ĵ�ı���Һ�����
���й������㣬���ݲ����������ʼ������۲��������ݷ�Ӧ2S2O32-+I2�TS4O62-+2I-����֪n��S2O32-��=2n��I2��������������Ʒ�IJ��ʣ�
��7��Na2S2O3��ԭ�Խ�ǿ������Һ���ױ�Cl2������SO42-����Ӧ���������ơ��Ȼ��ƣ���ƽ��д���ӷ���ʽ��
��� �⣺��1�����������ˮ�����Ҵ�����������ڷ�Ӧǰ���Ҵ�ʪ����ʹ������ڷ�ɢ����Һ�У�
�ʴ�Ϊ��ʹ������ڷ�ɢ����Һ�У�
��2������ͼʾ�������Ľṹ��������֪����aΪ�����ܣ���ʵ���������ܾ����������������ã�
�ʴ�Ϊ�������ܣ�����������
��3������Na2S2O3��Na2SO3���л�ԭ�ԣ��ױ���������Na2SO4�����Կ��ܴ��ڵ�������Na2SO4�����������Ƶķ���Ϊ��ȡ������Ʒ���ڹ���ϡ���ᣬ���ˣ�����Һ�м�BaCl2��Һ�����а�ɫ���������Ʒ�к���Na2SO4��
�ʴ�Ϊ��Na2SO4�� ȡ������Ʒ���ڹ���ϡ���ᣬ���ˣ�����Һ�м�BaCl2��Һ�����а�ɫ���������Ʒ�к���Na2SO4��
��4��S2O32?�������ӷ���������ԭ��Ӧ���ɵ���ɫ���ʣ���Ӧ�����ӷ���ʽΪ��S2O32?+2H+=S��+SO2��+H2O��
�ʴ�Ϊ��S2O32?+2H+=S��+SO2��+H2O��
��5���ζ������ⵥ��ʹ���۱��������Եζ��յ�ʱ��Һ��ɫ�仯Ϊ������ɫ��Ϊ��ɫ��
�ʴ�Ϊ������ɫ��Ϊ��ɫ��
��6������ͼʾ�ĵζ�����Һ���֪���ζ����г�ʼ����Ϊ0���ζ��յ�Һ�����Ϊ18.10mL���������ĵ�ı���Һ���Ϊ18.10mL��
20g Na2SO3����Բ����ƿ�У��ټ���80mL����ˮ����ȡ
4g�����ȫ��Ӧ��Ҫ����������Ϊ4g��$\frac{142}{32}$=15.75gg��С��20g���������ƹ�����������������Na2S2O3•5H2O�����ʵ���Ϊ$\frac{4g}{32g/mol}$=0.125mol��
���ݷ�Ӧ2S2O32-+I2�TS4O62-+2I-��֪��n��S2O32-��=2n��I2��=0.1000 mol•L-1��18.10��10-3L��2��$\frac{500mL}{20mL}$=9.05��10-2mol���ʲ���Ϊ��9.05��10-2mol��0.125mol����100%=72.4%��
�ʴ�Ϊ��72.4%��
��7��Na2S2O3��ԭ�Խ�ǿ������Һ���ױ�Cl2������SO42-����ƽ������ӷ���ʽΪ��S2O32?+4Cl2+5H2O=2SO42?+8Cl?+10H+��
�ʴ�Ϊ��S2O32?+4Cl2+5H2O=2SO42?+8Cl?+10H+��
���� ���⿼�黯ѧʵ���Ʊ����漰����ʶ�����ӵļ��鷽������װ�÷������ۡ�������ԭ��Ӧ�ζ�����㡢���ӷ���ʽ��д�ȣ��Ƕ�ѧ���ۺ������Ŀ��飬�Ѷ��еȣ�

 ��һ����ͬ���ɽ�����ϵ�д�
��һ����ͬ���ɽ�����ϵ�д� ������Ӧ���ϵ�д�
������Ӧ���ϵ�д� ��ʦ�㾦�ִʾ��ƪϵ�д�
��ʦ�㾦�ִʾ��ƪϵ�д�| A�� | ��ɢϵ�����ᡢ����ͭ������ | B�� | ����ʣ��ռ���ᡢ���� | ||
| C�� | �Σ��Ȼ��ơ���������̼��� | D�� | �ǵ���ʣ�ʯī��CO2������ |
| A�� | Ba2+��Ca2+��OH-��HCO3- | B�� | H+��K+��NH4+��SO42- | ||
| C�� | NO3-��SO42-��K+��Mg2+ | D�� | H+��Na+��NO3-��Cu2+ |
| A�� | һ����˫�ؽṹ���ķ���ʵ�������������� | |
| B�� | �÷���ԭ�Ӽ䶼�Թ��ۼ������� | |
| C�� | ��������һ���µĻ����� | |
| D�� | �����ʵ���Է�������Ϊ2400 |
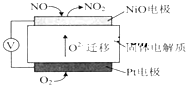 NOx������β���е���Ҫ��Ⱦ��֮һ��
NOx������β���е���Ҫ��Ⱦ��֮һ��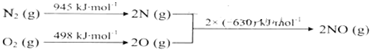
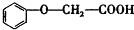 ����������Ӧ�IJ���A��һ��ʳ�����ϣ�����Ϊ��״�ṹ�Ҳ��������ش��������⣺
����������Ӧ�IJ���A��һ��ʳ�����ϣ�����Ϊ��״�ṹ�Ҳ��������ش��������⣺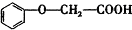 �ķ���ʽΪC8H8O3��
�ķ���ʽΪC8H8O3�� �ж���ͬ���칹�壬�����������࣬��ʹFeCl3��Һ����ɫ���ұ����ϵ�һ�ȴ��������ֵ�ͬ���칹��Ľṹ��ʽΪ
�ж���ͬ���칹�壬�����������࣬��ʹFeCl3��Һ����ɫ���ұ����ϵ�һ�ȴ��������ֵ�ͬ���칹��Ľṹ��ʽΪ ��
�� ��
�� ������һ�֣�����дһ�֣���
������һ�֣�����дһ�֣���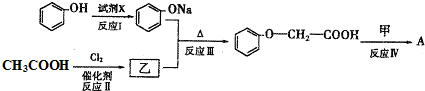
 ��
��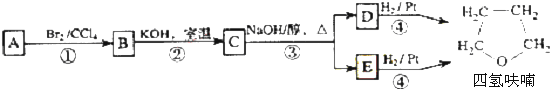
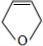 ��
��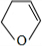 ��
��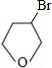 +NaOH$��_{��}^{H_{2}O}$
+NaOH$��_{��}^{H_{2}O}$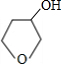 +NaBr��
+NaBr��