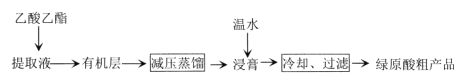
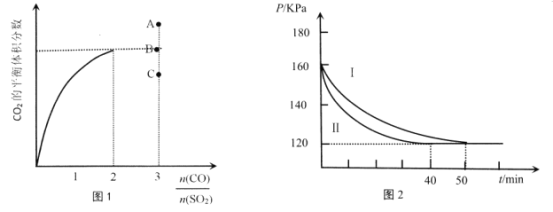
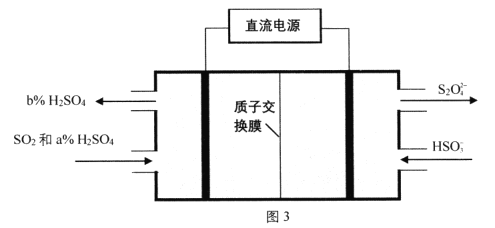
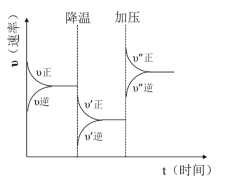
��Ŀ����
����Ŀ����ѧ��Ԥ��21������Ҷ�����������ܾ�����ʱ�����������ʶ��Ǿ��й���Ӧ��ǰ���Ĵ�����ϡ��ش��������⣺
(1)Zr(�)��Ԫ�����ڱ���λ�ڵ������ڣ�����ͬ�壬��̬Zr�ļ۲�����Ų�ʽΪ_______��
(2)�ǰ����(Li2NH) ����Ԫ�ص�һ��������С����____ ���縺��������_____ (��Ԫ�ط���)��
(3)����(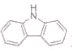 )�ķе����(
)�ķе����(![]() )�ߵ���Ҫԭ����________��
)�ߵ���Ҫԭ����________��
(4)��NH3BH3 (�����飬�۵�104��)�����黥Ϊ�ȵ����塣NH3BH3�ľ�������Ϊ____������B���ӻ�����Ϊ____����ͨ��_________�ⶨ�÷��ӵ����幹�͡�
��NH3BH3��ͨ�������顢CH4��H2O���кϳɣ����ǣ� CH4______H2O (����> "����<")��ԭ����________��
(5)MgH2�������ķ�Ʒϵ���ṹ��ͼ����������a =b= 450pm�� c= 30lpm��ԭ������ΪA(0��0��0)��B(0.305��0.305��0)��C(1��1��1)��D(0.195��0.805��0.5)��

��Mg2+�İ뾶Ϊ72pm����H+�İ뾶Ϊ______pm (�г��������ʽ)
����NA��ʾ�����ӵ�������MgH2����������ܶ��DZ�״���������ܶȵ�_____��(�г��������ʽ�������ܶ�Ϊ0.089g��L-1)��
���𰸡�4d25s2 Li N ������Ӽ������� ���Ӿ��� sp3 ������� �� CH4���µ��Ӷԣ�H2O�������Թµ��Ӷԣ��µ��Ӷ�������֮����ų��������� ��֮����ų�����ʹ��CH4�еļ��DZ�H2O �еļ��Ǵ� ![]()
![]() ��
�� ![]()
��������
��1��Zr(�)��Ԫ�����ڱ���λ�ڵ������ڣ�����ͬ�壬���ڢ�B�壬���̬Zr�ļ۲�����Ų�ʽΪ��
��2��Li��H����ͬ���壬��һ�������ɴ��С��H>Li���縺����С���H<Li ��Li��N����ͬ���ڣ���һ��������С���Li<N�� �縺����С���Li<N��
��3����������ӽṹ��ʽ��֪����������Ӽ����N��H��������۷е�ϸߣ�
��4����NH3BH3�۵�ϵͣ���NH3BH3Ϊ���Ӿ��壻��������ԭ�Ӽ۲���ӶԻ��������ж�B���ӻ����ͣ���ͨ��������ײⶨ�÷��ӵ����幹�ͣ�
��5�������ÿռ伸�μ���AB�ľ���=Mg2+�İ뾶+H+�İ뾶�������þ�̯�����㾧�������ܶȡ�
��1��Zr(�)��Ԫ�����ڱ���λ�ڵ������ڣ�����ͬ�壬���ڢ�B�壬���ݺ�������Ų�����д����̬Zr�ļ۲�����Ų�ʽΪ4d25s2���ʴ�Ϊ��4d25s2��
��2��Li��H����ͬ���壬��һ�������ɴ��С��H>Li���縺����С���H<Li ��Li��N����ͬ���ڣ���һ��������С���Li<N�� �縺����С���Li<N���ʴ�Ϊ��Li��N��
��3����������ӽṹ��ʽ��֪����������Ӽ����N��H��������۷е�ϸߣ��ʴ�Ϊ��������Ӽ���������
��4����NH3BH3�۵�ϵͣ���NH3BH3Ϊ���Ӿ��壻NH3BH3��Bԭ�Ӻ���3���Ҽ���1�Թµ��Ӷԣ����ӻ�����Ϊsp3�ӻ�����ͨ��������ײⶨ�÷��ӵ����幹�ͣ��ʴ�Ϊ��sp3��������ף�
������ԭ�ӹµ��Ӷ�Խ�����ԽС��CH4��H2O������sp3�ӻ���CH4���µ��Ӷԣ�H2O�������Թµ��Ӷԣ��µ��Ӷ�������֮����ų��������� ��֮����ų�����ʹ��CH4�еļ��DZ�H2O �еļ��Ǵʴ�Ϊ������CH4���µ��Ӷԣ�H2O�������Թµ��Ӷԣ��µ��Ӷ�������֮����ų��������� ��֮����ų�����ʹ��CH4�еļ��DZ�H2O�еļ��Ǵ�
��5�������ÿռ伸�μ���AB�ľ���=![]() =Mg2+�İ뾶+H+�İ뾶����H+�İ뾶=
=Mg2+�İ뾶+H+�İ뾶����H+�İ뾶=![]() ���ھ�����H+����Ϊ
���ھ�����H+����Ϊ![]() ����������ܶ�
����������ܶ�![]()
![]() ��
�� ![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
�� ![]() ��
�� ![]() ��
��

 ���Ǽ���С����ϵ�д�
���Ǽ���С����ϵ�д� �Ͻ�ƽ���Ȿϵ�д�
�Ͻ�ƽ���Ȿϵ�д�