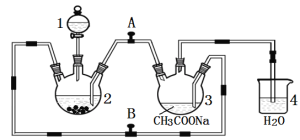
��Ŀ����
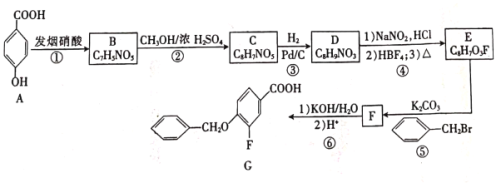
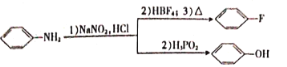
����Ŀ���ҹ��о���Ա���ֲ�Ҷ�е�EGCG����Ч����¹ڲ���S����������ACE2����Ľ�ϡ��Ʊ�EGCG���м���G��һ�ֺϳ�·�����£�
��֪��
�ش��������⣺
(1)G�й����ŵ�������______��
(2)��Ӧ�ٵķ�Ӧ������____________��
(3)F��KOH��Һ��Ӧ�Ļ�ѧ����ʽΪ__________________��
(4)�����廯����X��D��Ϊͬ���칹�壬��������������(![]() )��X��______�֣��Һ˴Ź���������3��壬�����֮��Ϊ6��2��1�Ľṹ��ʽΪ______(ֻдһ��)��
)��X��______�֣��Һ˴Ź���������3��壬�����֮��Ϊ6��2��1�Ľṹ��ʽΪ______(ֻдһ��)��
(5)̼ԭ��������4����ͬ��ԭ�ӻ����ʱ����̼ԭ�ӳ�Ϊ����̼�����й���EGCG(�ṹ��ʽ��ͼ)��˵���������______(����)��

A.�������ֹ�����
B.�����к�����������̼ԭ��
C.����![]() ��Һ������ɫ��Ӧ
��Һ������ɫ��Ӧ
D.1 mol EGCG��NaOH��Һ��Ӧ���������8 mol NaOH
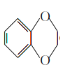
(6)1,4������������(![]() )���Ʊ�ԭ���Ը�Ѫѹҩ���ɳ��ີ��м��壬����Ա��Ӻ�1,2����������Ϊԭ���Ʊ�1,4������������ĺϳ�·��(���Լ�����)____________________��
)���Ʊ�ԭ���Ը�Ѫѹҩ���ɳ��ີ��м��壬����Ա��Ӻ�1,2����������Ϊԭ���Ʊ�1,4������������ĺϳ�·��(���Լ�����)____________________��
���𰸡��Ѽ����Ȼ�����ԭ�� ȡ����Ӧ 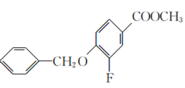 KOH
KOH 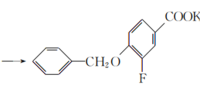 +CH3OH 14
+CH3OH 14  ��
�� AD
AD 
��������
����A��B�ķ���ʽ��֪��A�뷢�����ᷢ��ȡ����Ӧ��A�б����ϵ�һ����ԭ�ӱ�������ȡ��������B��C�ķ���ʽ��֪��B��״�����������Ӧ����C��C������������ԭ��Ӧ����D���������߷���ʽ��֪��C�����������������������֪����֪D����E�ķ�Ӧ��������֪��ͬ��D�а�����Fԭ�������E����ȡ����Ӧ����F��F����ˮ�ⷴӦ����G������G�Ľṹ��ʽ��֪��F�Ľṹ��ʽΪ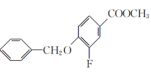 ��E�Ľṹ��ʽΪ
��E�Ľṹ��ʽΪ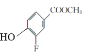 ��D�Ľṹ��ʽΪ
��D�Ľṹ��ʽΪ ��C�Ľṹ��ʽΪ
��C�Ľṹ��ʽΪ ��
��
(1)����G�Ľṹ��ʽ��֪��G�й����ŵ��������Ѽ����Ȼ�����ԭ�ӡ�
(2)���Ϸ�������Ӧ�ٵķ�Ӧ������ȡ����Ӧ��
(3)F�к�����������KOH��Һ����ˮ�ⷴӦ����ѧ����ʽΪ +KOH
+KOH  +CH3OH��
+CH3OH��
(4)D�ķ���ʽΪC8H9NO3�������廯����X��D��Ϊͬ���칹�壬��������������ȡ����-ONO2��-CH2CH3�����ڼ��3�ֽṹ����������������ȡ����-CH2ONO2��-CH3�����ڼ��3�ֽṹ������������3��ȡ����������-CH3��- ONO2������6�ֽṹ������������1��ȡ����-CH2CH2ONO2��ֻ��1�ֽṹ������������1��ȡ����-CH(CH3)ONO��ֻ��1�ֽṹ����X����������(![]() )�Ľṹ��14�֣����к˴Ź���������3��壬�����֮��Ϊ6��2��1�Ľṹ��ʽΪ
)�Ľṹ��14�֣����к˴Ź���������3��壬�����֮��Ϊ6��2��1�Ľṹ��ʽΪ ��
��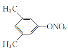 ��
��
(5)A.�������ǻ����������Ѽ����ֹ����ţ�A����
B.̼ԭ��������4����ͬ��ԭ�ӻ����ʱ����̼ԭ�ӳ�Ϊ����̼���ʷ����к�����������̼ԭ�� ��B��ȷ��
��B��ȷ��
C.�����б����������ǻ������ڷ�������![]() ��Һ������ɫ��Ӧ��C��ȷ��
��Һ������ɫ��Ӧ��C��ȷ��
D.1 mol EGCG�к���8mol���ǻ���1mol���������ǻ�����������NaOH��Һ��Ӧ���������9 mo NaOH��D����
��ѡAD��
(6)�Ա��Ӻ�1,2����������Ϊԭ���Ʊ�1,4�����������飬���� ��1,2���������鷴Ӧ�Ʊ�Ŀ�������ڱ��ӵ��ǻ���λ����һ���ǻ�����ͨ�������뷢�����ᡢ�������������Ƽ�H3PO2��Ӧ�����µ��ǻ�������ϳ�·�����£�
��1,2���������鷴Ӧ�Ʊ�Ŀ�������ڱ��ӵ��ǻ���λ����һ���ǻ�����ͨ�������뷢�����ᡢ�������������Ƽ�H3PO2��Ӧ�����µ��ǻ�������ϳ�·�����£�
 ��
��

����Ŀ����п��ZnS���������������Լ���ӫ���塢���ģ�����ʪ�������ױ�����ΪZnSO4���ش��������⣺
��1��д����̬Znԭ�ӵļ۵����Ų�ʽ__����̬Sԭ�Ӻ���δ�ɶԵ�����Ϊ__��
��2��ZnSO4������Ԫ�صĵ縺���ɴ�С��˳��Ϊ__��SO42-�����幹��Ϊ__������S���ӻ��������Ϊ__��
��3������п���ڰ�ˮ������[Zn(NH3)4]SO4��Һ��[Zn(NH3)4]SO4��Һ�в����ڵ�������������__��
a.���Ӽ� b.���ۼ� c.��λ�� d.���»��� e.���
��4����������п±������۵���ܽ��ԣ�����ZnCl2��ZnBr2��ZnI2�۵����������ԭ��__��
ZnF2 | ZnCl2 | ZnBr2 | ZnI2 | |
�۵�/�� | 872 | 275 | 394 | 446 |
���Ҵ����������ܽ��� | ���� | �ܽ� | �ܽ� | �ܽ� |
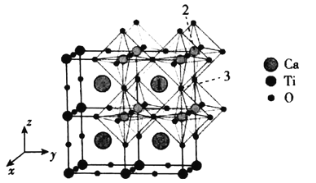
��5���ؾ��徧���ṹ��ͼ��ʾ�����������������ԭ�Ӿ���Ϊacm�������ӵ�����ΪNA��
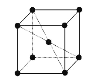
�ټ�ԭ�Ӻ����˶�״̬��ͬ�ĵ�������___��
�ڼؾ�����ܶ���___��