��Ŀ����
9����������һ�����Ϳ����オ��ĸ߷��Ӳ��ϣ���Ҫ��������ɽ�����ά���ɽ������Ϻ�ҽ�ò��ϣ���������Ϸ�������ˮ������ᣨ �������������������·ֽ�ΪCO2��H2O��
�������������������·ֽ�ΪCO2��H2O����1����ͼ�ף�����Ĺ�ҵ�������Բ�������ͷ���
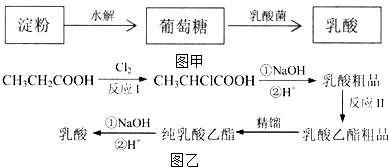
�ټ�������Ƿ���ȫˮ�⣬����ѡ�õ�һ���Լ���c������ţ���
a����ˮ b��������Һ c����� d���⻯�ص�����Һ
����18�������Ƿ��ͣ������Ͽ��Եõ������������18�֣�
��2����ͼ�ң����������Ҳ���Բ����л��ϳɷ������е�һ�ַ����DZ��ᷨ��
�ٷ�ӦI�ķ�Ӧ������ȡ����Ӧ��
�ڽ��з�ӦIIʱ����Ҫ����������Ҵ�����������Ŀ������������ת���ʣ�
��3�������������֮������γɻ�״�������û�״���Ľṹ��ʽ��
 ������һ�������·�����Ӧ�õ�������Ļ�ѧ����ʽ��n
������һ�������·�����Ӧ�õ�������Ļ�ѧ����ʽ��n $\stackrel{һ������}{��}$
$\stackrel{һ������}{��}$ +��n-1��H2O��
+��n-1��H2O��
���� ��1���ٵ�������ȫˮ�⣬��Һ��û�е���ʣ�࣬�����ƣ���Һ��������
���ɹ�ϵʽC6H12O6�������ǣ���2C3H6O3�����ᣩ����������غ㶨�ɣ���֪�������������������������
��2���������ʵĽṹ��Clԭ��ȡ����ΪHԭ�ӣ�
�����������Ҵ���ʹƽ������������ķ����ƶ���
��3������������Ӧԭ�����������ǻ�����������������ˮ���������ͨ��������Ӧ���е����۷�Ӧ���ɾ����ᣮ
��� �⣺��1���ٵ�������ȫˮ�⣬��Һ��û�е���ʣ�࣬�����ƣ���Һ����������ѡ��c��
���ɹ�ϵʽC6H12O6�������ǣ���2C3H6O3�����ᣩ����������غ㶨�ɣ���֪m�����ᣩ=m�������ǣ�=18�֣��ʴ�Ϊ��18��
��2���������ʵĽṹ����ӦI�DZ����Ц�ΪHԭ�ӱ�Clԭ��ȡ��������ȡ����Ӧ���ʴ�Ϊ��ȡ����
�����������Ҵ���ʹƽ������������ķ����ƶ����������ת���ʣ��ʴ�Ϊ�����������Ҵ���ʹƽ������������ķ����ƶ����������ת���ʣ�
��3������������Ӧԭ�����������ǻ�����������������ˮ���������������֮������γɻ�״�������û�״���Ľṹ��ʽ�� ��������һ�������·�����Ӧ�õ�������Ļ�ѧ����ʽ�ǣ�n
��������һ�������·�����Ӧ�õ�������Ļ�ѧ����ʽ�ǣ�n $\stackrel{һ������}{��}$
$\stackrel{һ������}{��}$ +��n-1��H2O��
+��n-1��H2O��
�ʴ�Ϊ�� ��n
��n $\stackrel{һ������}{��}$
$\stackrel{һ������}{��}$ +��n-1��H2O��
+��n-1��H2O��
���� ���⿼��������Ʊ����ϳ������ʣ����չ����ŵ�������ת���ǹؼ����Ѷ��еȣ�ע��Ի���֪ʶ���������գ�

| A�� | ʵ��������0.20 mol•L-1��NaOH��Һ950mL������ʱӦ��ȡNaOH������8.0g | |
| B�� | ����NH4Cl��Һ�¶ȣ���ˮ�����ӻ�������pH������ | |
| C�� | ˮ�������ȶ���ԭ����ˮ���Ӽ������� | |
| D�� | ��pH����2�Ĵ�����Һ�м�������0.01 mol•L-1��ϡ���ᣬ��Һ��pH���С |

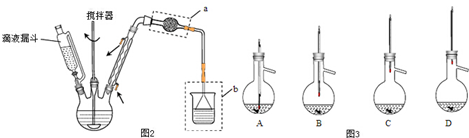
������þ���Ʊ����Ʊ�������ͼ1��ʾ��
��1����ͼ2��ʾװ�ý������գ�����A��������������
��2��ij�о�С��Ϊ���о�Mg2��OH��2CO3���������Ժϳ�MgO2��Ӱ�죮���ʵ�����±���ʾ����ȡ����������Ϊag����������������ʱ��̽�������¶ȶԺϳ�MgO2��Ӱ�죬��ѡ��ʵ��٢ۣ���ʵ���ţ���
| ʵ���� | �����¶ȣ��棩 | ����ʱ�䣨h�� |
| �� | 550 | 2 |
| �� | 600 | 1 |
| �� | 650 | 2 |
| �� | 700 | 3 |

��3��ij�о�С������ͼ3װ�òⶨһ����������Ʒ�й�����þ�ĺ�����
��ʵ��ǰ����еIJ����Ǽ��װ�õ������ԣ�ϡ�����м�������FeCl3��Һ������������������
���ú�ѹ��Һ©�����ŵ��У�����������Һ��������������������Ӱ�죻ʹ��Һ©���е���Һ˳�����£�
��4��ʵ���һ���ͨ�����з����ⶨ��Ʒ�й�����þ�ĺ�����
��ȡ1.600g��Ʒ����ƿ�У�����15mL����ˮ������ϡ���ᣬ��0.5000mol•L-1 KMnO4����Һ�ζ����յ㣮��Ӧ�����ӷ���ʽΪ��2MnO4-+5H2O2+6H+�T2Mn2++5O2��+8H2O
�ٵζ��յ�۲쵽������Ϊ���������һ��KMnO4��Һ����Һ����ɫ��Ϊdz��ɫ���Ұ�����ڲ���ɫ��
�ڸ���ͼ4�����Ʒ�е���������Ϊ89.25%��
| M | |
| T |
 ��Ԫ��W�����ڱ��е�λ���ǵ������ڵڢ�A�壮
��Ԫ��W�����ڱ��е�λ���ǵ������ڵڢ�A�壮M����̬�⻯��������������ˮ�������γ�һ�����ӻ���������ӻ�����Ļ�ѧʽΪNH4NO3��
��2���ڼ��������£�L�ĵ�����T������������ˮ�����Ũ��Һ��Ӧ�Ļ�ѧ����ʽΪC+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CO2��+2SO2��+2H2O��
L�ĵ������û���R�ĵ��ʣ���д����Ӧ�ķ���ʽ2C+SiO2$\frac{\underline{\;����\;}}{\;}$Si+2CO��
��3�������й���ӦԪ�طǽ�����ǿ���Ƚϵ�˵����ȷ����AC������ĸ��
A��M����̬�⻯���R����̬�⻯���ȶ�����ǽ�����Mǿ��R
B�������£�L�ĵ����ܴ�R��������������û���R����ǽ�����Lǿ��R
C��W�ĺ˵������T�࣬ԭ�Ӱ뾶��TС���õ�������ǿ����ǽ�����Wǿ��T��
| A�� | ��ȡþ����ˮ$\stackrel{Ca��OH��_{2}}{��}$Mg��OH��2$\stackrel{����}{��}$MgO$\stackrel{��ȼ}{��}$Mg | |
| B�� | ұ������������$\stackrel{����NaOH��aq��}{��}$NaAlO2$\stackrel{����HCl��aq��}{��}$��ˮAlCl3$\stackrel{���}{��}$Al | |
| C�� | ���壺Ũ����ˮ$\stackrel{Cl_{2}}{��}$Br2$��_{��ˮ}^{����}$$\stackrel{SO_{2}����}{��}$HBr��aq��$��_{��2���ٽ�һ������}^{��1��Cl_{2}}$Br2 | |
| D�� | �Ƶ⣺����$\stackrel{O_{2+}����}{��}$������$\stackrel{H_{2}O��ȡ}{��}$$\stackrel{Cl_{2}}{��}$I2��aq��$��_{��2���ٽ�һ������}^{��1���������ͣ���ȡ}$I2 |
| A�� | ʯ����ɵõ����͡�ú�͵� | |
| B�� | �ƻ���ϩ������C=C���������յ��������ƻ����������C-C������������ | |
| C�� | ��֬���Ǹ߷��ӻ����1mol��֬��ȫ��������1mol���ͺ�3 mol��֬���� | |
| D�� |  ��CO2��Ӧ���ɿɽ���ۺ��� ��CO2��Ӧ���ɿɽ���ۺ���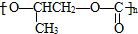 ���÷�Ӧ������ɫ��ѧ��ԭ�� ���÷�Ӧ������ɫ��ѧ��ԭ�� |


 ��
��