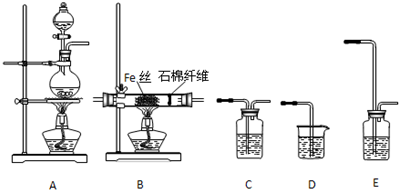
��Ŀ����
1����ҵ���Ʊ�������ʣ��漰�ķ�Ӧԭ�����������̽�Ϊ�������ǣ�������| A�� | ��ȡþ����ˮ$\stackrel{Ca��OH��_{2}}{��}$Mg��OH��2$\stackrel{����}{��}$MgO$\stackrel{��ȼ}{��}$Mg | |
| B�� | ұ������������$\stackrel{����NaOH��aq��}{��}$NaAlO2$\stackrel{����HCl��aq��}{��}$��ˮAlCl3$\stackrel{���}{��}$Al | |
| C�� | ���壺Ũ����ˮ$\stackrel{Cl_{2}}{��}$Br2$��_{��ˮ}^{����}$$\stackrel{SO_{2}����}{��}$HBr��aq��$��_{��2���ٽ�һ������}^{��1��Cl_{2}}$Br2 | |
| D�� | �Ƶ⣺����$\stackrel{O_{2+}����}{��}$������$\stackrel{H_{2}O��ȡ}{��}$$\stackrel{Cl_{2}}{��}$I2��aq��$��_{��2���ٽ�һ������}^{��1���������ͣ���ȡ}$I2 |
���� A��ȱ�ٹ��˲��裬���ɵ�������þ������Ҫ���˺��ܽ⣬�Ʊ�����þ��Ҫ����Ȼ�þ�õ���
B���������Ʊ���Ҫ������ڵ�����������ʯ�����壬�Ȼ����ǹ��ۻ����
C����ˮŨ���������������������������Ϊ�嵥�ʣ��������������պ�����ͨ�����������廯��õ��嵥�ʣ�
D���������ͺ��в����������͵ⵥ�ʻᷢ���ӳɷ�Ӧ��
��� �⣺A��ȱ�ٹ��˲��裬���ɵ�������þ������Ҫ���˺��ܽ⣬�Ʊ�����þ����Ҫ������ڵ��Ȼ�þ�õ������ǵ������þ����A����
B��������ڵ�����������ʯ�������Ʊ����������Ȼ����ǹ��ۻ�������ڵ��Ȼ������ܵ��磬��B����
C����ˮŨ���������������������������Ϊ�嵥�ʣ��������������պ�����ͨ�����������廯��õ��嵥�ʣ������ʵ�������ȷ����C��ȷ��
D���������ͺ��в����������͵ⵥ�ʻᷢ���ӳɷ�Ӧ����������ȡ����Ӧ�÷������ͣ���D����
��ѡC��
���� ���⿼���˺�ˮ��Դ�ķ���Ӧ�ã�������ұ��������ԭ�������ջ�����ҵ���������ǽ���ؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
7��������������ķ�����һ����ȵ��ǣ�������
| A�� | ������ȡ��ܶȲ��ȵ�N2��C2H4 | B�� | ��ͬѹǿ����ͬ�����CO��O2 | ||
| C�� | ��ͬ�¶ȡ���ͬ�����O2��N2 | D�� | ������ȵ�N2��CO2 |
16����֪25��ʱ��
��HF��aq��+OH-��aq��?F-��aq��+H2O��1����H=-67.7kJ/mol
��H+��aq��+OH-��aq���TH2O��1����H=-57.3kJ/mol
�����й�˵������ȷ���ǣ�������
��HF��aq��+OH-��aq��?F-��aq��+H2O��1����H=-67.7kJ/mol
��H+��aq��+OH-��aq���TH2O��1����H=-57.3kJ/mol
�����й�˵������ȷ���ǣ�������
| A�� | HF�������� | |
| B�� | ˮ������0.1 mol Fʱ�����յ�����Ϊ6.77 kJ | |
| C�� | �к��Ⱦ�Ϊ57.3 kJ/mol | |
| D�� | ��0.1 mol HF��0.1 mol NaOH��������Һ��Ϻ�ų�������Ϊ6.77 kJ |
6�����в��ֶ�����Ԫ�ص����ʻ�ԭ�ӽṹ���±���
��1��д��AԪ�������ڱ��е�λ�õ������ڵڢ�A�壬BԪ�ص�����̼��
��2��д��CԪ�صĵ��ʵĵ���ʽ ��
��
��3��Ԫ��D��Ԫ��A��ȣ��ǽ����Խ�ǿ����Cl����Ԫ�ط��ű�ʾ�������б�������֤����һ��ʵ����b����ѡ����ţ���
a��������D�ĵ��ʺ�A�ĵ���״̬��ͬ
b��D���⻯���A���⻯���ȶ�
c��һ��������D��A�ĵ��ʶ������Ʒ�Ӧ
��4��̽Ѱ���ʵ����ʲ�������ѧϰ����Ҫ����֮һ��A��B��C��D����Ԫ�ص���������� ��ˮ�����л�ѧ�������Բ�ͬ���������ֵ���H2CO3��д��ѧʽ����
| Ԫ�ر�� | Ԫ�����ʻ�ԭ�ӽṹ |
| A | ���������Ӳ㣬��K��M�������֮�͵���L������� |
| B | �����������Ǵ������2�� |
| C | �����µ���Ϊ˫ԭ�ӷ��ӣ����⻯���ˮ��Һ�ʼ��� |
| D | Ԫ�����������+7�� |
��2��д��CԪ�صĵ��ʵĵ���ʽ
 ��
����3��Ԫ��D��Ԫ��A��ȣ��ǽ����Խ�ǿ����Cl����Ԫ�ط��ű�ʾ�������б�������֤����һ��ʵ����b����ѡ����ţ���
a��������D�ĵ��ʺ�A�ĵ���״̬��ͬ
b��D���⻯���A���⻯���ȶ�
c��һ��������D��A�ĵ��ʶ������Ʒ�Ӧ
��4��̽Ѱ���ʵ����ʲ�������ѧϰ����Ҫ����֮һ��A��B��C��D����Ԫ�ص���������� ��ˮ�����л�ѧ�������Բ�ͬ���������ֵ���H2CO3��д��ѧʽ����
10��NAΪ�����ӵ���������ֵ������˵������ȷ���ǣ�������
| A�� | ���ܱ������м���l.5mol H2��0.5molN2����ַ�Ӧ��ɵõ�NH3������ΪNA | |
| B�� | һ�������£�2.3g��Na��ȫ��O2��Ӧ����3.6g����ʱʧȥ�ĵ�����Ϊ0.1NA | |
| C�� | ���³�ѹ�£�2.24LCO��CO2��������к��е�̼ԭ����ĿΪ0.1NA | |
| D�� | ��״���£�22.4L��CCl4�к��е�CCl4������ΪNA |
 �������������������·ֽ�ΪCO2��H2O��
�������������������·ֽ�ΪCO2��H2O��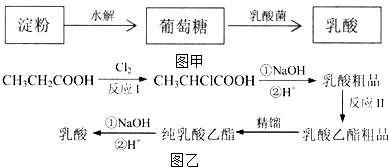
 ������һ�������·�����Ӧ�õ�������Ļ�ѧ����ʽ��n
������һ�������·�����Ӧ�õ�������Ļ�ѧ����ʽ��n $\stackrel{һ������}{��}$
$\stackrel{һ������}{��}$ +��n-1��H2O��
+��n-1��H2O��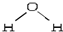 �������ͽṹ��������ɵ����IJ����غϣ����Ϊ���Է��ӣ�CO2�ĽṹʽΪ O�TC�TO������ ��ɵ������غϣ�Ϊ�Ǽ��Է��ӣ���֪���Է��������ڼ��Է����У����Ǽ��Է��������ڷǼ��Է����У�����������ԭ����
�������ͽṹ��������ɵ����IJ����غϣ����Ϊ���Է��ӣ�CO2�ĽṹʽΪ O�TC�TO������ ��ɵ������غϣ�Ϊ�Ǽ��Է��ӣ���֪���Է��������ڼ��Է����У����Ǽ��Է��������ڷǼ��Է����У�����������ԭ����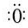 ��
�� ��
��