��Ŀ����
���к͵ζ����ⶨij�ռ���Ʒ�Ĵ���,�Ը���ʵ��ش���������:
��1��ȷ����8.2 g�������������������ʵ���Ʒ,���500 mL������Һ?����ʱ,��Ʒ�ɷ�
��________��������ĸ������?
A��С�ձ��� B���ྻֽƬ�� C��������
��2�� �ζ�ʱ,��0.2000 mol��L-1���������ζ�������Һ,����ѡ��____��������ĸ����ָʾ��?
A������ B��ʯ�� C����̪
��3��������̨�ϵ�һ�Ű�ֽ,��Ŀ����_______ _?
��4����0.20 mol��L-1���������ζ�10.00 mL������Һ���ζ���ֹʱ����������Һ20.00mL�����㱻���ռ���Һ�����ʵ���Ũ����________mol��L-1,�ռ���Ʒ�Ĵ�����________?
��5������ʵ�����������ƿ�ô���Һ��ϴ,Ȼ���ټ���10.00 mL����Һ,��ζ���� ���ƫ�ߡ�?��ƫ�͡�����Ӱ�족����
��1��A��1�֣�
��2��B ��1�֣�
��3�����ڹ۲���ƿ��Һ����ɫ�ı仯,��С�ζ���2�֣�
��4��0.40��2�֣� 97.56% ��2�֣�
��5��ƫ�ߣ�1�֣�
���������������1���ռ����ǿ��ʴ�ԣ�����ʱӦ����С�ձ��н��У�ѡA.��
��2�� ���ζ��Ĺؼ����ǵζ��յ���жϣ��ζ��յ������ָʾ����ָʾ��Ϊ�˸��õ��жϵζ��յ㣬���Dz����ܹ���dzɫ������ɫ��ָʾ��������Ҫ������ɫ����dzɫ��ָʾ��������ָʾ���ı�ɫ��ΧԽխԽ�á����ȵı�ɫ��Χ��3.1��4.4��ʯ��ı�ɫ��Χ��5��8����̪�ı�ɫ��Χ��8��10��������ζ��ռ���Һʱ��һ��ѡ����ȣ�Ҳ��ѡ���̪��������ѡ��ʯ�� ����ѡ B��
��3��������̨�ϵ�һ�Ű�ֽ��Ŀ���DZ��ڹ۲���ƿ�ڵ���ɫ�仯����С��
��4����������к͵ζ���ԭ�����ɼ��������Һ��Ũ��Ϊ��0.2mol/L��0.02L/0.01L=0.4mol/L�����ռ�����Ϊ0.4mol/L��0.5L��40g/mol=8g,������Ʒ�Ĵ���Ϊ8g/8.2g��100%=97.56%��
��5������ƿ�ô���Һ��ϴ��ʵ�����������˴���Һ�е����ʵ��������ñ�Һ���ƫ�ߣ�ʹʵ����ƫ�ߡ�
���㣺�����к͵ζ��IJ�����ָʾ����ѡ���㣬������

ijѧ����0.200 0 mol��L��1�ı�NaOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ�����Ϊ���¼�����
��������ˮϴ�Ӽ�ʽ�ζ��ܣ�������ע��NaOH��Һ����0���̶�������
�ڹ̶��õζ��ܲ�ʹ�ζ��ܼ������Һ��
�۵���Һ������0����0���̶�������ijһ�̶ȣ������¶���
����ȡ20.00 mL����Һע��ྻ����ƿ�У�������3�η�̪��Һ
���ñ�Һ�ζ����յ㣬���µζ���Һ�����
��ش�
(1)���ϲ����д������(����) ���ô�������ᵼ�²ⶨ���(�ƫ����ƫС������Ӱ�족) ��
(2)�жϵζ��յ�������ǣ���ƿ����Һ�� ɫ��Ϊ ɫ���Ұ�����ڲ���ɫ��
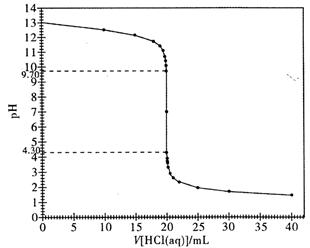
(3)��ͼ��ij�εζ�ʱ�ĵζ����е�Һ�棬�����Ϊ mL��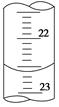
(4)�����������ݣ���������������Һ��Ũ��Ϊ mol/L
| �ζ����� | ����Һ���(mL) | ��NaOH��Һ������¼(mL) | |
| �ζ�ǰ���� | �ζ������ | ||
| ��һ�� | 20.00 | 0.40 | 20.40 |
| �ڶ��� | 20.00 | 4.00 | 24.00 |
| ������ | 20.00 | 2.00 | 24.10 |
����������������Ʒ��������̨������Ȧ����ʽ���У� ����ƿ ����ʽ�ζ��ܺͼ�ʽ�ζ��� ���ձ������ɣ� �ݲ����� ��ͷ�ι� ����ƽ�������룩 ����ֽ ����Ͳ �����©����
����ҩƷ��
| A��NaOH���� | B����NaOH��Һ | C��δ֪Ũ������ | D������ˮ��E��̼������Һ |
��1������ʱ��Ӧѡ�õ����������� �����ţ���
��2������250mlһ�����ʵ���Ũ�ȵ�NaOH��Һʱ����ȱ�ٵ������� ��
��3��������к͵ζ�ʱ����ȱ�����Լ��� ��
��4�������к͵ζ�ʱ���������ý�Ҫʢ����Һ������ϴ�����������е� �����ţ���
�ף���ʽ�ζ��� �ң���ʽ�ζ��� ����25 mL��Ͳ ������ƿ
��5��ijͬѧ��һ����֪Ũ�ȵ�������Һ�ζ�δ֪Ũ�ȵ�NaOH��Һ�����в����Եζ��Ľ����ʲôӰ�죿���ƫ����ƫС������Ӱ�족��
����ʽ�ζ���������ˮϴ����δ�ñ�������ϴ��_____________
�ڵζ�ǰ���ζ��ܼ�������ݣ��ζ���������ʧ��_____________
�۶���ʽ�ζ��ܵĿ̶�ʱ���ζ�ǰ���Ӱ�Һ����ʹ����ζ����Ӷ�����_______
�ܵζ����յ㸽��ʱ������ƿ�м�����������ˮ��ϴƿ����մ����Һ_____
��ͼ��ʾ�ĵ���I��II�У�a��b��c��d��ΪPt�缫���������У��缫b��d��û�������ݳ���������������������b>d����������ʵ����������Һ��( )
| ѡ�� | X | Y |
| A�� | AgNO3 | Cu(NO3)2 |
| B�� | MgSO4 | CuSO4 |
| C�� | FeSO4 | Al2 (SO4)3 |
| D�� | CuSO4 | AgNO3 |
��ͼ��ʾ��a��b�Ƕ��ʯī�缫��ijͬѧ��ͼʾװ�ý�������ʵ�飺 �Ͽ�K2���պ�K1һ��ʱ�䣬�۲쵽��֧�������ڶ������ݽ��缫��Χ����ʱ�Ͽ�K1���պ�K2���۲쵽������A��ָ����ƫת������˵������ȷ����
| A���Ͽ�K2���պ�K1һ��ʱ�䣬��Һ��pH��� |
| B���Ͽ�K1���պ�K2ʱ��b���ϵĵ缫��ӦʽΪ��2H++2e��====H2�� |
| C���Ͽ�K2���պ�K1ʱ��a���ϵĵ缫��ӦʽΪ��4OH����4e��====O2��+2H2O |
| D���Ͽ�K1���պ�K2ʱ��OH����b���ƶ� |

 H++H2PO3����
H++H2PO3����