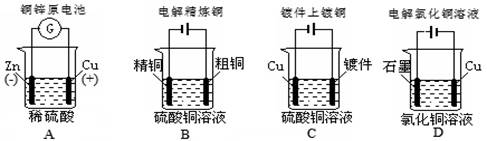
��Ŀ����
����������������Ʒ��������̨������Ȧ����ʽ���У� ����ƿ ����ʽ�ζ��ܺͼ�ʽ�ζ��� ���ձ������ɣ� �ݲ����� ��ͷ�ι� ����ƽ�������룩 ����ֽ ����Ͳ �����©����
����ҩƷ��
| A��NaOH���� | B����NaOH��Һ | C��δ֪Ũ������ | D������ˮ��E��̼������Һ |
��1������ʱ��Ӧѡ�õ����������� �����ţ���
��2������250mlһ�����ʵ���Ũ�ȵ�NaOH��Һʱ����ȱ�ٵ������� ��
��3��������к͵ζ�ʱ����ȱ�����Լ��� ��
��4�������к͵ζ�ʱ���������ý�Ҫʢ����Һ������ϴ�����������е� �����ţ���
�ף���ʽ�ζ��� �ң���ʽ�ζ��� ����25 mL��Ͳ ������ƿ
��5��ijͬѧ��һ����֪Ũ�ȵ�������Һ�ζ�δ֪Ũ�ȵ�NaOH��Һ�����в����Եζ��Ľ����ʲôӰ�죿���ƫ����ƫС������Ӱ�족��
����ʽ�ζ���������ˮϴ����δ�ñ�������ϴ��_____________
�ڵζ�ǰ���ζ��ܼ�������ݣ��ζ���������ʧ��_____________
�۶���ʽ�ζ��ܵĿ̶�ʱ���ζ�ǰ���Ӱ�Һ����ʹ����ζ����Ӷ�����_______
�ܵζ����յ㸽��ʱ������ƿ�м�����������ˮ��ϴƿ����մ����Һ_____
��1���٢ܢݢ�⡣��2��250ml������ƿ��
��3�����ָʾ��(��̪�����)��
��4������5����ƫ���ƫ���ƫС����Ӱ��
���������������1��������Ҫ�õ������У�����̨������Ȧ����ʽ���У����ձ�������������ֽ������©����
��2������250mlһ�����ʵ���Ũ�ȵ�NaOH��Һ��Ҫ250mL����ƿ��
��3����ȷ���жϵζ��յ���Լ������ָʾ����
��4�������ϴ��ƿ����ʹ��ƿ����ȡ��Һ���࣬�����
��5������ʽ�ζ���δ�ñ�������ϴ����ʹ�������Ũ�ȱ�С����ɵζ����ƫ��
�ڵζ�ǰ���ζ��ܼ�������ݣ��ζ���������ʧ��˵���������������ֵ����ʵ��ֵ�����ƫ��
�۵ζ�ǰ���Ӱ�Һ����ʹ����ζ����Ӷ�������Һ�IJ���ֵ��С�����ƫС��
������ƿ�м�����������ˮ��ϴƿ����մ����Һ����Ӱ���������������Եζ������Ӱ�졣
���㣺���⿼�����������ѡ������һ�����ʵ���Ũ����Һ���ۺϵζ���

 ���ʿ��ÿ��ֳɳ�ϵ�д�
���ʿ��ÿ��ֳɳ�ϵ�д�ij��ѧʵ��С����Ҫ�˽��г�����ʳ�ð״ף���Ҫ�Ǵ����ˮ��Һ����ȷŨ�ȣ��ִ��г�������һƿijƷ��ʳ�ð״ף���ʵ���ұ�NaOH��Һ������еζ���
��1����ʵ��Ӧѡ�� ��ָʾ������ȡһ������İ״����õ������� ��
��2����ͼ��ʾ50mL�ζ�����Һ���λ�ã���A��C�̶ȼ����1mL��A���Ŀ̶�Ϊ25���ζ�����Һ�����ӦΪ mL��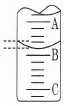
��3��Ϊ�˼�Сʵ������ͬѧһ��������3��ʵ�飬����ÿ����ȡ�״������ΪVmL��NaOH��ҺŨ��Ϊcmo1/L��3��ʵ������¼���£�
| ʵ����� | ��һ�� | �ڶ��� | ������ |
| ����NaOH��Һ���/mL | 26.02 | 25.35 | 25.30 |
���ϱ����Կ�������һ��ʵ���м�¼����NaOH��Һ��������Զ��ں����Σ���ԭ������ǣ� ��
A��������ȷ��ʵ�����ʱ���ӿ̶��߶�ȡ�ζ��յ�ʱNaOH��Һ�����
B���ζ�ǰ�ζ��ܼ��������ݣ��ζ�����������
C��ʢװ��Һ�ĵζ���װҺǰ������ˮϴ����δ�ñ�Һ��ϴ
D���μ�NaOH��Һ���죬δ������տ�����Һ��ɫ������ֹͣ�ζ�
��4���״���Һ�д��ڵĵ���ƽ��Ϊ ���õ���ƽ�ⷽ��ʽ��ʾ����
��5���ڵζ������У���c(CH3COO��)>c(Na+)ʱ��������Һ��____������ţ���
A������ B������ C������ D����ȷ��
��6�������������ݣ�д������ð״��д�������ʵ���Ũ�ȵı���ʽ�����ػ���c= ��


 I��+ I2��ʵ���ҿ���ͨ��������ԭ�ζ����ⶨƽ��ʱI3����Ũ��
I��+ I2��ʵ���ҿ���ͨ��������ԭ�ζ����ⶨƽ��ʱI3����Ũ��