��Ŀ����
����Ŀ���о���Ԫ�ؼ��仯��������ʾ�����Ҫ���塣
(1)25 �棬��0.10 mol��L��1H2S��Һ�У�ͨ��HCl��������NaOH�����Ե�����ҺpH����ҺpH��c(S2��)��ϵ����ͼ(������Һ����ı仯��H2S�Ļӷ�)��

��pH��11ʱ����Һ�е�c(H2S)��c(HS��)��________mol��L��1��
��ij��Һ��0.020 mol��L��1Mn2����0.10 mol��L��1H2S������ҺpH��________ʱ��Mn2����ʼ������[��֪��Ksp(MnS)��2.8��10��13]
(2)25 �棬������ĵ��볣�����±���
Ka1 | Ka2 | |
H2SO3 | 1.3��10��2 | 6.3��10��8 |
H2CO3 | 4.2��10��7 | 5.6��10��11 |
��HCO![]() �ĵ��볣������ʽK��________��
�ĵ��볣������ʽK��________��
��0.10 mol��L��1Na2SO3��Һ������Ũ���ɴ�С��˳��Ϊ___________________��
��H2SO3��Һ��NaHCO3��Һ��Ӧ����Ҫ���ӷ���ʽΪ__________________________��
���𰸡� 0.087 5 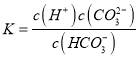 c(Na��)>c(SO
c(Na��)>c(SO![]() )>c(OH��)>c(HSO
)>c(OH��)>c(HSO![]() )>c(H��) H2SO3��HCO
)>c(H��) H2SO3��HCO![]() ===HSO
===HSO![]() ��H2O��CO2��
��H2O��CO2��
��������(1) �� ����SԪ�������غ���c(H2S)��c(HS��)+c(S2-)��0.1 mol��L��1 �� pH��11ʱ��c(S2-)=0.013mol/L������c(H2S)��c(HS��)=0.1mol/L-0.013mol/L=0.087mol/L���ڵ���Һ��Mn2+��Ũ����S2-Ũ�ȵ�Ũ����Qc> Ksp(MnS)ʱ��Mn2+��ʼ������������0.020��c(S 2-)>2.8��10��13��c(S2-)>1.4��10-11������ͼ���֪��ʱpH��5����pH��5ʱMn2+ ��ʼ������
(2) �ٸ��ݶ���д��HCO3-�ĵ��볣������ʽ: 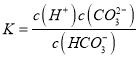 ����Na2SO3��Һ�У�Na+��Ũ�����SO32-������������ˮ�⣬��һ��ˮ����ڵڶ���ˮ�⣬����ˮ�������OH-������ˮ��õ���OH-��Ũ�ȴ���HSO3����Ũ�ȣ�H+Ũ����С����������Ũ���ɴ�С��˳��Ϊ��c(Na��)>c(SO32-)>c(OH��)>c(HSO3-)>c(H��)���۸���������ĵ��볣����֪H2SO3�����Դ���H2CO3�����ԣ�����H2SO3��NaHCO3��Ӧʱ�ܹ�����H2CO3�����ӷ���ʽΪ��H2SO3+HCO3-=HSO3-+CO2��+H2O��
����Na2SO3��Һ�У�Na+��Ũ�����SO32-������������ˮ�⣬��һ��ˮ����ڵڶ���ˮ�⣬����ˮ�������OH-������ˮ��õ���OH-��Ũ�ȴ���HSO3����Ũ�ȣ�H+Ũ����С����������Ũ���ɴ�С��˳��Ϊ��c(Na��)>c(SO32-)>c(OH��)>c(HSO3-)>c(H��)���۸���������ĵ��볣����֪H2SO3�����Դ���H2CO3�����ԣ�����H2SO3��NaHCO3��Ӧʱ�ܹ�����H2CO3�����ӷ���ʽΪ��H2SO3+HCO3-=HSO3-+CO2��+H2O��

 �ƸԴ��ž�ϵ�д�
�ƸԴ��ž�ϵ�д�����Ŀ���������ػ������ڻ�����ҽҩ�����ϵ��������Ź㷺��Ӧ�á��ش�����������
(1)��̬Asԭ�ӵĺ�������Ų�ʽΪ[Ar]_________��Asԭ�ӵ�������(kJ/mol)����������
��һ������ | �ڶ������� | ���������� | ���ĵ����� | ��������� | ���������� |
947.0 | 1798 | 2735 | 4837 | 6043 | 12310 |
�����������������������������ԭ��:_____________________________________��
(2)�����Ǿ����۷��ӣ����ͽṹ����֤�������ǷǾ������ɿ�������__________��
A.���� B.ԭ�ӷ������ C.�˴Ź����� D.X��������
(3)���������Ͷ�ά�뵼����ϣ���������ʯīһ����Ƭ��ṹ(��ͼ)�������֮����_____��ϡ��ӽṹ�Ͽ���������ϩ����������ʯīϩ��ԭ����____________________________��

(4)����(P4)��������������ṹ��3.1g��������������ĿΪ________������(P4)�����ڶ���̼��������ˮ����ԭ����_____________________________________________��
(5)GaAs���۵�Ϊ1238�棬�侧���ṹ��ͼ��ʾ���þ��������Ϊ_________��Gaԭ�ӵ��ӻ���ʽΪ______________��ÿ��Asԭ����Χ�����Asԭ����ĿΪ________����֪GaAs���ܶ�Ϊdg/cm3��Ħ������ΪMg/mol�����������As��Gaԭ�Ӻ˼��Ϊ_____(�����ӵ�����ֵ��NA��ʾ���г���ʽ����)nm��

����Ŀ��̼����������ɫ���壬������ˮ����һ����Ҫ�Ĺ�ҵԭ�ϣ��������Ʊ���Ѫ������������Ҳ�������ɳ���صĵ缫��ij�о�С��ͨ������ʵ�飬Ѱ�����ø��ֽⷴӦ�Ʊ� FeCO3 ��������ѷ�����
ʵ�� | �Լ� | ���� | |
�ι� | �Թ� | ||
| 0.8 mol/L FeSO4 ��Һ��pH=4.5�� | 1 mol/L Na2CO3 ��Һ ��pH=11.9�� | ʵ���������������ɫ������5min ��������Եĺ��ɫ |
0.8 mol/L FeSO4 ��Һ��pH=4.5�� | 1 mol/L NaHCO3 ��Һ ��pH=8.6�� | ʵ�������ɫ������������ɫ���ݣ�2min ��������ԵĻ���ɫ | |
0.8 mol/L (NH4)2Fe(SO4)2 ��Һ��pH=4.0�� | 1 mol/L NaHCO3 ��Һ ��pH=8.6�� | ʵ�������ɫ��������ɫ���ݣ��ϳ�ʱ�䱣�ְ�ɫ | |
(1)ʵ�� I �к��ɫ����������ԭ��������·�Ӧ��ʾ���벹ȫ��Ӧ��
��Fe2+ +��____+�� + ��H2O=�� Fe(OH)3 +��HCO3
(2)ʵ�� II �в��� FeCO3 �����ӷ���ʽΪ_____________________________��
(3)Ϊ��̽��ʵ�� III �� NH4+��������ã���ͬѧ�����ʵ�� IV ����̽����
���� | ���� | |
ʵ�� IV | �� 0.8 mol/L FeSO4 ��Һ�м����__________���ټ��� Na2SO4 �������Ƴɻ����Һ����֪ Na+��ʵ����Ӱ�죬���Ի�Ϻ���Һ����仯������ȡ����Һһ�ιܣ��� 2mL 1 mol/L NaHCO3 ��Һ��� | ��ʵ�� III ������ͬ |
ʵ�� IV �м��� Na2SO4 �����Ŀ���Ǣ�_______________________��
�Ա�ʵ�� II��III��IV����ͬѧ�ó����ۣ�NH4+ˮ����� H+��������Һ pH�������˸����� Fe(OH)2 �IJ�����
��ͬѧ��Ϊ��ʵ�鷽�������Ͻ���Ӧ����ĶԱ�ʵ������ǣ���_____________����ȡ����Һһ�ιܣ��� 2mL 1 mol/L NaHCO3 ��Һ��ϡ�
(4)С��ͬѧ��һ��������Ϊ������ʵ��������ֱ��֤��ʵ�� III �� FeCO3 �Ĵ�����ߣ���Ҫ������ͼ��ʾ��װ�ý��ж����ⶨ���ֱ�ʵ�� I��II��III �еij������й��ˡ�ϴ�ӡ�����������Ȼ��ת���� A ���Ĺ��ƿ�С�

Ϊ�ⶨ FeCO3 �Ĵ��ȣ�����Ʒ�������⣬����ⶨ����������______________________��
(5)ʵ�鷴˼�����ⶨ��ʵ�� III �е� FeCO3 ���ȸ��ڷ��� I �ͷ��� II��ͨ������ʵ��������Ʊ� FeCO3 ʵ��ɹ��Ĺؼ�������_______________________________________