��Ŀ����
Χ�������������ʣ���������A12O3����ϡ���ᣬ��H2SO4����Ba(OH)2���壬�ް�ˮ����A12(SO4)3������Ҫ��ش��������⡣
��1��������ǿ�ᷴӦ��������ǿ�Ӧ���� ������ţ���
��2�����ڵ���ʵ��� ������ţ���
��3��д����������ȡAl(OH)3�����ӷ���ʽ ��
��4�������ʷ�����Ӧ�����ӷ���ʽΪH++OH��=H2O����д���÷�Ӧ�Ļ�ѧ����ʽ ��
��5��34.2g������ˮ���500mL��Һ����Һ��SO42�������ʵ���Ũ��Ϊ ��
��6������۷�����Ӧ�Ļ�ѧ����ʽΪA1+4HNO3=A1(NO3)3+NO��+2H2O���÷�Ӧ�л�ԭ���������������ʵ���֮���� ��
��1���٢ڣ���1�֣����1����0.5�֣����һ�������֣�
��2���ڢܢݢߣ���2�֣����1����0.5�֣����һ�������֣�
��3��Al3����3NH3��H2O=Al(OH)3����3NH4������2�֣�
��4��2HNO3+Ba(OH)2= Ba(NO3)2+2H2O����2�֣�
��5��0.6mol/L����2�֣���λδд��0.5�֣�
��6��1��1����1�֣�
���������������1��Al��Al2O3������ǿ�ᷴӦ��������ǿ�Ӧ����2���������ᡢ��Ρ��������������ڵ���ʣ����ʡ�����Һ������Һ���ǵ���ʣ���3��ʵ���ҳ���������Һ�백ˮ��ȡAl(OH)3��Al3����3NH3��H2O=Al(OH)3����3NH4������4��H++OH��=H2O����ǿ����ǿ�������ɿ������κ�ˮ���кͷ�Ӧ��2HNO3+Ba(OH)2= Ba(NO3)2+2H2O����5����m/M��֪��n[A12(SO4)3]=0.1mol����A12(SO4)3��3SO42����֪��n(SO42��)=0.3mol����n/V��֪��c(SO42��)=0.6mol/L����6����Ԫ��ȫ����0�����ߵ�+3�ۣ�����������Al�ǻ�ԭ����1/4��Ԫ����+5�۽��͵�+2�ۣ�����ԭ����HNO3������������÷�Ӧ�л�ԭ����������֮��Ϊ1��1��
���㣺������Ԫ�ػ��������ʡ����ӷ���ʽ�����ʵ��������ʵ���Ũ�ȡ���ԭ���������������֪ʶ��

��֪Ca(OH)2��Cl2��Ӧ�������������¶��йأ���һ������ʯ������ͨ��һ����������������ǡ����ȫ��Ӧ(�����ķ�Ӧ��Ϊ���ȷ�Ӧ)���������к���Cl����ClO����ClO3�����ֺ���Ԫ�ص����ӣ�����ClO����ClO3���������ӵ����ʵ���(n)�뷴Ӧʱ��(t)��������ͼ��ʾ��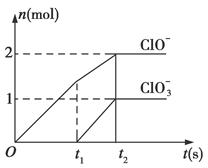
(1)t1ǰ������������______________________(�ѧʽ)��
(2)t2ʱ��Ca(OH)2��Cl2������Ӧ���ܵ����ӷ���ʽ��_____________________________��
(3)��ʯ�����к���Ca(OH)2�����ʵ�����________mol��
(4)NaClO2���ȶ��������Ȼ��û��������ƹ���ʱ������ը���䱬ը��IJ��������________(����ĸ)��
| A��NaCl��Cl2 | B��NaCl��NaClO | C��NaClO3��NaClO4 | D��NaCl��NaClO3 |
ijУͬѧΪ̽��Br2��I2��Fe3����������ǿ��������������ʵ�顣
ʵ��٣�ȡ����KI��Һ���Թ��У��ȼ�����ˮ�����ټ���CCl4�����ã��۲쵽�²�Һ����Ϻ�ɫ��
ʵ��ڣ�ȡ����FeSO4��Һ���Թ��У��ȼ�����ˮ�����ټ����μ�����KSCN��Һ�����۲쵽��Һ�ʺ�ɫ��
��1��д�����ӷ���ʽ��
ʵ��٣�______________________________________________________��
ʵ��ڣ�______________________________________________________��
��2������������ʵ�飬�����ʵ������Կ��Եó�����ȷ������________��
| A��Br2>I2 | B��Fe3��>Br2 | C��Br2>Fe3�� | D��I��>Br�� |
��FeCl3��Һ���ڵ�ˮ����KI��Һ����ϡH2SO4���ݵ�����Һ
ij��Һ�н����±������е�5������(������ˮ�ĵ��뼰���ӵ�ˮ��)���Ҹ����ӵ����ʵ�����Ϊ0.1mol��
| ������ | SO42����NO3����Cl�� |
| ������ | Fe3����Fe2����NH4+��Cu2����Al3�� |
������ԭ��Һ�м���KSCN��Һ������������
������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䣻
������ԭ��Һ�м���BaCl2��Һ���а�ɫ�������ɡ�
�Իش��������⣺
��1������ԭ��Һ���ȼ����������ᣬ�ٵ���KSCN��Һ��ʵ��������________________��ʵ��������������Ӧ�����ӷ���ʽ��_________________________��
��2��ԭ��Һ��������������(д���ӷ���)___________________.
��3������ԭ��Һ�м�������NaOH��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������________________g��
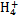 ��Fe2+��Fe3+��C
��Fe2+��Fe3+��C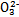 ��S
��S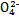 ��Cl-��N
��Cl-��N �е�һ�ֻ���,ȡ����Һ����ʵ��,ʵ����������:
�е�һ�ֻ���,ȡ����Һ����ʵ��,ʵ����������: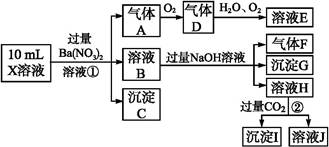
 Al(OH)4��+ H+ ���� Cl2+2OH��= Cl��+ ClO��+H2O
Al(OH)4��+ H+ ���� Cl2+2OH��= Cl��+ ClO��+H2O