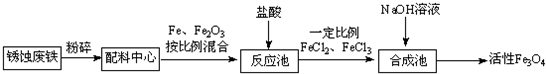
��Ŀ����
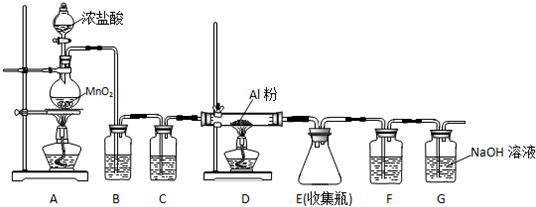
16��ij��ȤС��������ͼװ���Ʊ�����þ��̽������þ��ijЩ���ʣ�����ɺ����ϵ�������⣺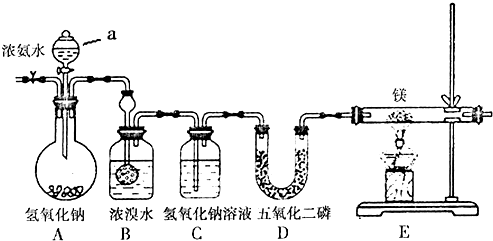
��1��ʵ�鲽�輰����
��������������������ԣ�Ȼ����������м�����������ҩƷ��
��ͨ��������壬�ų�װ���ڵĿ�����
�۴�Һ©���������۲쵽B��Ũ��ˮ��ɫ��dz����ɫ��C�в�����ɫ���ݣ�
�ܵ�ȼ�ƾ��ƣ��۲쵽E�й������˻���ɫ��������þ�ǻ���ɫ���壬��ˮ���ҷ�Ӧ�������ּ��
��2��Aװ��������a������Ϊ��Һ©����
��3��д��B����Ӧ�Ļ�ѧ����ʽ3Br2+8NH3�T6NH4Br+N2��
��4��Dװ���е�P2O5��һ�ַ������Ը�������������岻����Ũ����������P2O5�������b��
a��NH3 b��HI C��SO2 d��CO2
��5��Eװ���з�����Ӧ�Ļ�ѧ����ʽΪ��N2+3Mg$\frac{\underline{\;����\;}}{\;}$Mg3N2��
��6����װ���д���������ȱ�ݣ���ĸĽ���ʩ�ǣ���Eװ�ú�����װ�м�ʯ�ҵĸ���ܣ���ֹ�����е�ˮ��������Eװ�ã�
��7��ijͬѧҪ̽��E�й���ɷ֣���E�еĹ������ϡ�����У��۲쵽�����ܽⲢ�������ݣ�д���˹����з�Ӧ�Ļ�ѧ����ʽMg3N2+8HCl�T2MgCl2+2NH4Cl��Mg+2HCl�TMgCl2+H2����
���� �������Ʊ�����þ��ʵ������⣬������������������ȡ���������������������õ�����������������Һ���ӷ����壬���������������ﵪ��������ڼ��ȵ������þ�뵪����Ӧ���ɵ���þ��
��1��������װ���漰��������Ʊ��������Ҫ����װ�������ԣ�
����þ��Ҳ����������Ӧ�������Ҫ�ṩ�������������⸱��������ɣ�
��2��װ��A��a�����ǵμ�Ũ��ˮ�ģ��Ƿ�Һ©����
��3��B�����������������Ƶ�����ͬʱ�����廯泥�
��4���ɸ���Ũ������ǿ���Լ�ǿ�����ԣ�������������ˮ��Ӧ��������������ж�
��5��E����þ���ڵ�����ȼ�����ɵ���þ��
��6����þ����ˮ�⣬���Eװ�ú�Ҫ��һ������װ�ã��������е�ˮ�����룻
��7������þ����ˮ�⣬���ɰ�����������þ��ˮ������ٺ�ϡ���ᷴӦ�������л��й�����þҲ�ܺ����ᷴӦ��
��� �⣺��1�����Ʊ������װ�ã���Ҫ����װ�������ԣ��ʴ�Ϊ�����װ�������ԣ�
��Ϊ��ֹþ����������Ӧ�������Ҫͨ��������壬�ų�װ���еĿ������ṩ�������������⸱��������ɣ��ʴ�Ϊ��ͨ��������壬�ų�װ���еĿ�����
��2��װ��A��a�����Ƿ�Һ©�����ʴ�Ϊ����Һ©����
��3��B�з�����Ӧ�Ļ�ѧ����ʽΪ3Br2+8NH3�T6NH4Br+N2���ʴ�Ϊ��3Br2+8NH3�T6NH4Br+N2��
��4��NH3�����ᷴӦ��HI����ǿ��ԭ�ԣ��ʵ���������Ũ������Ҳ�������������������HI������Ũ���������������������CO2��SO2�������ö��߸���ʴ�Ϊ��b��
��5��E�з�����Ӧ�Ļ�ѧ����ʽΪN2+3Mg$\frac{\underline{\;����\;}}{\;}$Mg3N2���ʴ�Ϊ��N2+3Mg$\frac{\underline{\;����\;}}{\;}$Mg3N2��
��6��Ϊ��ֹ����þˮ�⣬��Eװ�ú�Ҫ��һ������װ�ã��������е�ˮ�����룬�ʴ�Ϊ����Eװ�ú�����װ�м�ʯ�ҵĸ���ܣ���ֹ�����е�ˮ��������Eװ�ã�
��7���ɿ��ɵ���þ��ˮ���ɰ�����������þ���ٺ����������Ӧ�����������þҲ�ܺ����ᷴӦ��������Ӧ�Ļ�ѧ����ʽΪMg3N2+8HCl�T2MgCl2+2NH4Cl��Mg+2HCl�TMgCl2+H2�����ʴ�Ϊ��Mg3N2+8HCl�T2MgCl2+2NH4Cl��Mg+2HCl�TMgCl2+H2����
���� ���⿼�鰱�����Ʊ������ʡ�þ�����ʼ�ʵ��Ļ����������ۺ��ԱȽ�ǿ�����ѶȽϵͣ��ص㿼��������������


| A�� | W��ԭ������������Y������ | |
| B�� | Z��ԭ�Ӱ뾶��X�Ĵ� | |
| C�� | YԪ�صķǽ����Ա�ZԪ�ص�ǿ | |
| D�� | Z������������Ӧ��ˮ��������Ա�W��ǿ |
A������ʳ��ˮ B��ˮ C��ŨH2SO4 D����ʯ�� E������ F���������� G��Ũ���� H���ռ���Һ
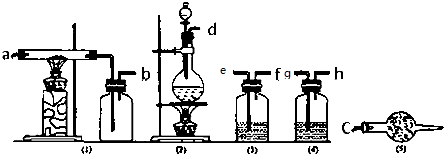
�ش��������⣺��1��д��AlCl3¶���ڿ��������ɰ����Ļ�ѧ����ʽ��AlCl3+3H2O?Al��OH��3+3HCl��
��2��������������ѡȡ��������һ�Ʊ���������ˮAlCl3��װ�ã���ͼ�и��ܿڱ�Ű��Ⱥ������Ϊ��d��e��f��g��h��a��b��c��
��3����д����װ���и�ѡ��������Ӧʢ�ŵ����ʣ�
| ������� | ��1�� | ��2�����ϣ� | ��2�����£� | ��3�� | ��4�� | ��5�� |
| �Լ���� | G | F |

| A�� | �Ʊ���ʽ�����������˹�������������� | |
| B�� | Ϊ��ֹNH4HCO3�ֽ⣬����FeCO3���ڽϵ��¶��½��� | |
| C�� | ����KSCN��Һ���飨NH4��2Fe��SO4��2�Ƿ����� | |
| D�� | �Ʊ���NH4��2Fe��SO4��2�����������ܽ�ȱ�FeSO4���ܽ�ȴ���һ���� |
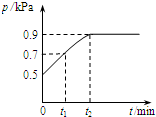 ��1.0L�ܱ������з���0.1mol X����һ���¶��·�����Ӧ��X��g��?Y��g��+Z��g����H��0 ������������ѹǿp�淴Ӧʱ��t�ı仯��ϵ��ͼ��ʾ�����·�����ȷ���ǣ�������
��1.0L�ܱ������з���0.1mol X����һ���¶��·�����Ӧ��X��g��?Y��g��+Z��g����H��0 ������������ѹǿp�淴Ӧʱ��t�ı仯��ϵ��ͼ��ʾ�����·�����ȷ���ǣ�������| A�� | t1ʱn��X��=0.04 mol | |
| B�� | t1��t2����������ƽ����Է��������� | |
| C�� | �����ƽ����ϵ��Y�ĺ�������������ϵ�¶Ȼ����Z���� | |
| D�� | �����������䣬�ٳ���0.1 mol ����X��ƽ�������ƶ���X��ת���ʼ��� |
| A�� | ��ΪSiO2��H2SiO3����������SiO2����ˮ��H2SiO3 | |
| B�� | CO2ͨ��ˮ������Na2SiO3��ˮ��Һ���п��Եõ�������� | |
| C�� | ��Ϊ����ʱ������Ӧ��Na2CO3+SiO2$\frac{\underline{\;����\;}}{\;}$Na2SiO3+CO2�������Թ�������Ա�̼��ǿ | |
| D�� | SiO2���������������������� |