��Ŀ����
����Ŀ��FeSO4��Һ�����ڿ��������ױ��ʣ����Ϊ�˷���ʹ��Fe2+��ʵ�����г�������������茶���[�׳ơ�Ħ���Ρ�����ѧʽΪ(NH4)2Fe(SO4)26H2O]�������̷����̷���Һ���ȶ���
I��ij��ȤС�����ʵ���Ʊ���������茶��塣
��ʵ���У�������Һ�Լ�����ʹ�õ�������ˮ��������С���ȴ����ʹ�á���FeSO4��Һ�м��뱥��(NH4)2SO4��Һ����������_______����ȴ�ᾧ�����ˡ�ϴ�Ӻ����õ�һ��dz����ɫ�ľ��塣
II��ʵ��̽��Ӱ����Һ��Fe2+�ȶ��Ե�����
(1)����0.8 mol/L��FeSO4��Һ��pH=4.5����0.8 mol/L��(NH4)2Fe(SO4)2��Һ��pH=4.0������ȡ2 mL������Һ����֧�Թ��У��տ�ʼ������Һ����dz��ɫ���ֱ�ͬʱ�μ�2��0.01mol/L��KSCN��Һ��15min��۲�ɼ���(NH4)2Fe(SO4)2��Һ��ȻΪdz��ɫ��������Һ��FeSO4��Һ����ֵ���ɫ���ǡ�
������1��
���� | Fe(OH)2 | Fe(OH)3 |
��ʼ���� pH | 7.6 | 2.7 |
��ȫ���� pH | 9.6 | 3.7 |
���������ӷ���ʽ����FeSO4��Һ��������ɫ���ǵ�ԭ��___________________��
������Ӱ��Fe2+�ȶ��Ե����أ�С��ͬѧ�������3�ּ��裺
����1������������ͬʱ��NH4+�Ĵ���ʹ(NH4)2Fe(SO4)2��Һ��Fe2+�ȶ��ԽϺá�
����2������������ͬʱ����һ�� pH��Χ�ڣ���Һ pHԽСFe2+�ȶ���Խ�á�
����3��__________________________________________________��
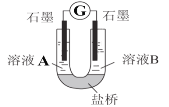
(2)С��ͬѧ����ͼװ�ã�GΪ���������ƣ�������������������Һ�ֱ������ҺA��0.2 mol/L NaCl������ҺB��0.1mol/L FeSO4��Ϊ��ͬ�� pH���۲��¼�����ƶ������Լ���2����ʵ���о���ʵ�������±���ʾ��
��� | A 0.2mol/LNaCl | B 0.1mol/LFeSO4 | �����ƶ��� |
ʵ��1 | pH=1 | pH=5 | 8.4 |
ʵ��2 | pH=1 | pH=1 | 6.5 |
ʵ��3 | pH=6 | pH=5 | 7.8 |
ʵ��4 | pH=6 | pH=1 | 5.5 |
������2��ԭ���װ���У�����������ͬʱ��������Ӧ��Ļ�ԭ��Խǿ��������Ӧ���������Խǿ����ԭ��صĵ���Խ��
������3�������£�0.1mol/LpH=1��FeSO4��Һ��pH=5��FeSO4��Һ�ȶ��Ը��á�
��������ʵ������������Ϣ����С�����ۿ��Եó����½��ۣ�
��U������صĵ缫��Ӧʽ_________________��
�ڶԱ�ʵ��1��2����3��4������һ��pH��Χ�ڣ��ɵó��Ľ���Ϊ______ ��
�۶Ա�ʵ��_____��_____ ���ɵó���һ�� pH��Χ�ڣ���Һ����Ա仯�Ƕ�O2������ǿ����Ӱ�����ء�
�ܶԣ�����3��ʵ����ʵ�Ľ���Ϊ____________________��
���𰸡�����Ũ�� 4Fe2+ + O2 + 10H2O��4Fe(OH)3�� + 8H+ ������������ͬʱ�����������Ũ�ȴ�СӰ��Fe2+���ȶ��ԣ�������������ͬʱ�����������Ũ��Խ��Fe2+���ȶ��ԽϺá��� O2 + 4e- +4H+��2H2O ��Һ����Խǿ��Fe2+�Ļ�ԭ��Խ�� 1 3����2��4�� ����������ͬʱ����Һ������ǿ��Fe2+�Ļ�ԭ�Լ�����Ӱ�죬�����˶�O2����������ǿ��Ӱ�죬��pH=1��FeSO4��Һ���ȶ�������������������֣�
��������
�����������ʼ�ʵ��������������Ļ�ѧ��Ӧ������������Һ����Ũ�ȵIJ����жϣ�NH4+��SO42-�Լ�H+����Ӱ��Fe2+�ȶ��ԣ��ɱ������ݿ�֪����Һ����Խǿ�������ƶ���ԽС��˵��Fe2+�Ļ�ԭ��Խ������ʵ��1��3����2��4����֪��pHԽ������ƶ���ԽС���Դ˽����⡣
I��FeSO4���л�ԭ�ԣ�Ӧ���ⱻ������������ˮ��������С���ȴ����ʹ�ã���ȥˮ���ܽ������������Һ�õ����壬Ӧ�ȼ���Ũ����Һ��Ȼ����ȴ�ᾧ�����ˡ�ϴ�Ӻ���ʴ�Ϊ������Ũ����
II����1����FeSO4��Һ���ֵ���ɫ���ǣ�Ӧ���������������������ӷ���ʽΪ4Fe2++O2+10H2O=4Fe��OH��3��+8H+���ʴ�Ϊ��4Fe2++O2+10H2O=4Fe��OH��3��+8H+��
�ڸ���������Һ����Ũ�ȵIJ����жϣ�NH4+��SO42-�Լ�H+����Ӱ��Fe2+�ȶ��ԣ��������Ϣ��֪������3��Ϊ������������ͬʱ�����������Ũ�ȴ�СӰ��Fe2+���ȶ��ԣ�������������ͬʱ�����������Ũ��Խ��Fe2+���ȶ��ԽϺã����ʴ�Ϊ��������������ͬʱ�����������Ũ�ȴ�СӰ��Fe2+���ȶ��ԣ�������������ͬʱ�����������Ũ��Խ��Fe2+���ȶ��ԽϺã���
��2����AΪ�Ȼ�����Һ����������������������Ӧ���缫����ʽΪO2+4e-+4H+=2H2O���ʴ�Ϊ��O2+4e-+4H+=2H2O��
���ɱ������ݿ�֪����Һ����Խǿ�������ƶ���ԽС��˵��Fe2+�Ļ�ԭ��Խ�����ʴ�Ϊ����Һ����Խǿ��Fe2+�Ļ�ԭ��Խ����
����ʵ��1��3����2��4����֪��pHԽ������ƶ���ԽС���ɵó���һ��pH��Χ�ڣ���Һ����Ա仯��O2������ǿ����Ӱ�����أ��ʴ�Ϊ��1��3����2��4����
���ɱ������ݿ�֪��pH�仯���������ӵĻ�ԭ�Խ�ǿ�������ƶ�������ϴ�֪����������ͬʱ����Һ������ǿ��Fe2+�Ļ�ԭ�Լ�����Ӱ�죬�����˶�O2����������ǿ��Ӱ�졣��pH=1��FeSO4��Һ���ȶ����ʴ�Ϊ������������ͬʱ����Һ������ǿ��Fe2+�Ļ�ԭ�Լ�����Ӱ�죬�����˶�O2����������ǿ��Ӱ�죬��pH=1��FeSO4��Һ���ȶ���

����Ŀ��Cu��Zn���仯������������������������Ҫ���á�
��1��Cu��Zn�����ڱ���__������ɫ��ӦʱCu��4s���ӻ�ԾǨ��4p�����д��Cu�ļ���̬�����Ų�ʽ_��
��2���ֱ���CuSO4��MgSO4��Һ�Ӱ�ˮ��������ǰ��Ϊ����ɫ��Һ������Ϊ��ɫ������
��NH3��Cu2+�γ���������������Mg2+��ԭ����__��
����Һ�е�ˮ����H3O+��H5O2+������ʽ��H5O2+�ɿ�����H3O+��H2Oͨ������γɵ����ӣ���H5O2+�Ľṹʽ��__��
��3��Zn��ij�ֻ�����M�ǺܺõIJ�п�����ṹʽ��ͼ��
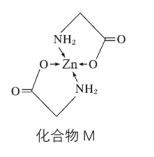
��1molM���е���������ĿΪ__��
�ڳ�����N��������H2NCH2COO-��NH3��N3-�ȣ�NH3�ķ��ӿռ乹��Ϊ__��N3-������Nԭ���ӻ���ʽΪ__��
��M�������������ʸߵ�ԭ�������п�γ���������__���������������������������������������������ά�����õ��ȶ��ԡ�
��4��±��п���۵������
±��п/ZnX2 | ZnF2 | ZnCl2 | ZnBr2 | ZnI2 |
�۵�/�� | 872 | 283 | 394 | 445 |
��ZnX2�۵�����仯��ԭ����__��
��5��Zn��ij������ľ�����ͼ��ʾ����֪�����ܶ�Ϊdgcm-3����S2-��Zn2+���У���Zn2+��S2+�ĺ˼��Ϊ__nm��д�������ʽ����

����Ŀ���±���Ԫ�����ڱ���һ���֡��밴Ҫ����գ�
IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 | |
1 | ||||||||
2 | �� | �� | �� | �� | �� | |||
3 | �� | �� | �� | �� | �� |
(1)���Т�~���Ԫ���У�ԭ�Ӱ뾶������________(���������)���ǽ�������ǿ��Ԫ����________(���������)������������Ӧ��ˮ������������ǿ����_______(���������)����ѧ�������ȶ�����________(���������)��
(2)Ԫ�آ۵ij����⻯��ĵ�ʽΪ_________�����⻯����Ԫ�آ���⻯�ﻯ�Ϻ�����ˮ��ˮ��Һ������Ũ���ɴ�С��˳����________(�����ӷ��ű�ʾ)��
(3)��Ԫ�آߵĵ�����ɰֽ��ĥ���ϼ���![]() ��ҺƬ�̺������γɰ�ɫë״���ë״��Ļ�ѧʽ��______________����Ԫ�آߵĵ��ʵķ�ĩ���������ڸ����·�Ӧ��ų������ȣ�д���÷�Ӧ�Ļ�ѧ����ʽ____________
��ҺƬ�̺������γɰ�ɫë״���ë״��Ļ�ѧʽ��______________����Ԫ�آߵĵ��ʵķ�ĩ���������ڸ����·�Ӧ��ų������ȣ�д���÷�Ӧ�Ļ�ѧ����ʽ____________