��Ŀ����
����Ŀ����FeO��Cu(NO3)2�Ļ����9.08gȫ������300mL��1mol/L�������У��в���Fe2+�����������µ�NO3-�������仯ѧ��Ӧ����ʽΪ��3Fe2+��NO3-��4H+��3Fe3+��NO����2H2O�����ų�NO����448mL����״��������������Һ��ͨ��һ������Cl2ǡ�ý���Һ�е�Fe2����ȫ�������ټ���10g���������ۣ���ַ�Ӧ�ų�H2�����ˣ��õ����ܹ��塣������������ȷ����
A��Cu(NO3)2�����ʵ���Ϊ0.01mol
B��ͨ��Cl2���������״����Ϊ448mL
C���ų�H2���������״����Ϊ112mL
D���õ����ܹ��������Ϊ6.72g
���𰸡�C
��������
�������������������ᷴӦ�����Һ��ͨ��һ����������ʱ����Һ�е�Fe2+�պñ���ȫ������������Һ�м���10g���������ۣ���ַ�Ӧ�������������˵������������ᷴӦʱ���������ȫ��Ӧ����������ʣ�࣬A��NO�����ʵ���=![]() =0.02mol�����ݵ�Ԫ���غ㣬��֪����ͭ���ʵ���=
=0.02mol�����ݵ�Ԫ���غ㣬��֪����ͭ���ʵ���= ![]() =0.01mol����A��ȷ��B��NO�����ʵ���=
=0.01mol����A��ȷ��B��NO�����ʵ���=![]() =0.02mol�����ݵ�Ԫ���غ㣬��֪����ͭ���ʵ���=
=0.02mol�����ݵ�Ԫ���غ㣬��֪����ͭ���ʵ���= ![]() =0.01mol��������ͭ����=0.01mol��188g/mol=1.88g������������������=9.08-1.88g=7.2g���������������ʵ���=
=0.01mol��������ͭ����=0.01mol��188g/mol=1.88g������������������=9.08-1.88g=7.2g���������������ʵ���=![]() =0.1mol����ͨ������Ϊx mol�����ݵ���ת���غ㣺0.1��1=0.02����5-2��+2x�����x=0.02mol����ͨ�����������Ϊ0.02mol��22.4L/mol=0.448L=448mL����B��ȷ��C������NO�����ʵ���Ϊ0.02mol�����ݷ���ʽ3FeCl2+HNO3+3HCl�T3FeCl3+NO��+2H2O����֪����HCl�����ʵ���=0.02mol����3+1��=0.08mol����FeO+2HCl=FeCl2+H2O��֪����HClΪ0.1mol��2=0.2mol���ʹ�����HClΪ0.28mol��ʣ��HClΪ0.3mol-0.28mol=0.02mol��������ԭ���غ㣬�������������ʵ���=
=0.1mol����ͨ������Ϊx mol�����ݵ���ת���غ㣺0.1��1=0.02����5-2��+2x�����x=0.02mol����ͨ�����������Ϊ0.02mol��22.4L/mol=0.448L=448mL����B��ȷ��C������NO�����ʵ���Ϊ0.02mol�����ݷ���ʽ3FeCl2+HNO3+3HCl�T3FeCl3+NO��+2H2O����֪����HCl�����ʵ���=0.02mol����3+1��=0.08mol����FeO+2HCl=FeCl2+H2O��֪����HClΪ0.1mol��2=0.2mol���ʹ�����HClΪ0.28mol��ʣ��HClΪ0.3mol-0.28mol=0.02mol��������ԭ���غ㣬�������������ʵ���=![]() =0.01mol�����������������=0.01mol��22.4L/mol=0.224L=224mL����C������D����FeԪ���غ��֪����Һ��Fe3+Ϊ0.1mol����Fe+2Fe3+=3Fe2+��֪������FeΪ
=0.01mol�����������������=0.01mol��22.4L/mol=0.224L=224mL����C������D����FeԪ���غ��֪����Һ��Fe3+Ϊ0.1mol����Fe+2Fe3+=3Fe2+��֪������FeΪ![]() =0.05mol����Һ��Cu2+Ϊ0.01mol����Fe+Cu2+=Cu+Fe2+��֪������FeΪ0.01mol����������Ϊ0.01mol����Fe+2H+=Fe2++H2������֪����FeΪ0.01mol�����ܹ�����Fe������=��0.05+0.01+0.01��mol��56g/mol=3.92g��ʣ��Fe������=10g-3.92g=6.08g������ܹ���ΪCu��ʣ��Fe������֮�ͣ��ʲ��ܹ�������=6.08g+0.01mol��64g/mol=6.72g����D��ȷ����ѡC��
=0.05mol����Һ��Cu2+Ϊ0.01mol����Fe+Cu2+=Cu+Fe2+��֪������FeΪ0.01mol����������Ϊ0.01mol����Fe+2H+=Fe2++H2������֪����FeΪ0.01mol�����ܹ�����Fe������=��0.05+0.01+0.01��mol��56g/mol=3.92g��ʣ��Fe������=10g-3.92g=6.08g������ܹ���ΪCu��ʣ��Fe������֮�ͣ��ʲ��ܹ�������=6.08g+0.01mol��64g/mol=6.72g����D��ȷ����ѡC��

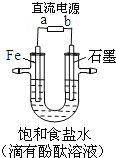
 �Ƹ�С״Ԫ�������������ϵ�д�
�Ƹ�С״Ԫ�������������ϵ�д� ����һ������ܼƻ�ϵ�д�
����һ������ܼƻ�ϵ�д�����Ŀ��PCl3��PCl5������Ҫ�Ļ���ԭ�ϡ���PCl3(g)��Cl2(g)������������2L�ܱ������У���һ�������·�����Ӧ��PCl3(g)��Cl2(g)![]() PCl5(g)������10minʱ�ﵽƽ�⡣�й��������£�
PCl5(g)������10minʱ�ﵽƽ�⡣�й��������£�
PCl3(g) | Cl2(g) | PCl5(g) | |
��ʼŨ��(mol��L��1) | 2.0 | 1.0 | 0 |
ƽ��Ũ��(mol��L��1) | c1 | c2 | 0.4 |
�����жϲ���ȷ����
A.10min�ڣ�v(Cl2)��0.04mol��L��1��min��1
B.��������Cl2Ϊ1.2molʱ����Ӧ�ﵽƽ��
C.�����¶�(T1��T2)����Ӧ��ƽ�ⳣ����С��ƽ��ʱPCl3��![]() ��1
��1
D.ƽ�������2.0molPCl3��1.0molCl2����ͬ�������ٴ�ƽ�⣬c(PCl5)��0.2mol��L��1