��Ŀ����
A��B��C��D��E 5�ֶ���������Ԫ�ط�ռ�������ڣ�A��B��CΪͬһ�����������ڵ�3��Ԫ�أ�A��C��ԭ������֮��Ϊ3��4��Eԭ�ӵĵ��Ӳ�������������������D��ԭ������С��E�����û�ѧ����ش�������⣺
��1��AԪ�������ڱ��е�λ�� ��
��2���Ƚ�C��E�����Ӱ뾶��С�� ��
��3��Ԫ��E��һ�ֳ����Ŀ���������Һ�ʼ��ԣ���ԭ���ǣ������ӷ���ʽ��ʾ����
��
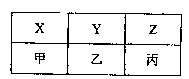 ��4��X��Y��Z���ס��ҡ�������A��B��C�ֱ���D��
��4��X��Y��Z���ס��ҡ�������A��B��C�ֱ���D��
�ɵĻ�������ֻ���������ų��±�������ͬһ
���еķ����е�������ͬ��ͬһ���е���������Ԫ
��������ͬ������X��Y���׳��³�ѹ��Ϊ���壬
Z���ҡ������³�ѹ��ΪҺ�塣
�ټķ���ʽΪ �����ĵ���ʽΪ ��
���Һͱ���������ƽ�����ȼ�ϣ���Ӧ��IJ�������Ⱦ����֪8gҺ̬��������Һ̬����ȫ��Ӧ�������Ϊ����ʱ���ų�����Ϊ160��35kJ����д���÷�Ӧ���Ȼ�ѧ����ʽ�� ��
��5��2007��7��2�գ�������������ѧ�ҳɹ��ϳ��˾��ж��ػ�ѧ���Ե�E2D6�����д������C2����ȫȼ�յĻ�ѧ����ʽ�� ��
��1���ڶ����ڢ�A�塣
��2��O2->Al3+��2�֣���
 ��3��A1O2-+2H2O
Al(OH)3+OH-��2�֣�
��3��A1O2-+2H2O
Al(OH)3+OH-��2�֣�
��4����C2H6��H2O2����ʽ��
��N2H4(1)+2H2O2(1)=N2(g)+4H2O(g),��H=-641.40kJ/mol��3�֣�
|
��5��A12H6+3O2 Al2O3+3H2O��3�֣�

 [��ѧ/ѡ��/���ʽṹ������]A��B��C��D��E���ֶ�����Ԫ�أ�ԭ��������������Ԫ�ض�Ӧ�ĵ��ʾ�Ϊ���壮A��C��E��Ԫ�ص�ԭ�Ӻ����ֻ��2��δ�ɶԵ��ӣ�B��EԪ�ص�ԭ������֮�͵���C��DԪ�ص�ԭ������֮�ͣ�
[��ѧ/ѡ��/���ʽṹ������]A��B��C��D��E���ֶ�����Ԫ�أ�ԭ��������������Ԫ�ض�Ӧ�ĵ��ʾ�Ϊ���壮A��C��E��Ԫ�ص�ԭ�Ӻ����ֻ��2��δ�ɶԵ��ӣ�B��EԪ�ص�ԭ������֮�͵���C��DԪ�ص�ԭ������֮�ͣ�