��Ŀ����
����Ŀ�����仯����Ӧ�ù㷺���ش��������⣺
��1����̬Bԭ�ӵļ۵��ӹ������ʽΪ________�����һ�����ܱ�Be ________�����С������
��2�������飨NH3BH3������Ϊ�����DZ�������ʹ������֮һ�������д�����λ�����ṩ�µ��ӶԵijɼ�ԭ����________��д��һ���백���黥Ϊ�ȵ�����ķ���________���ѧʽ����
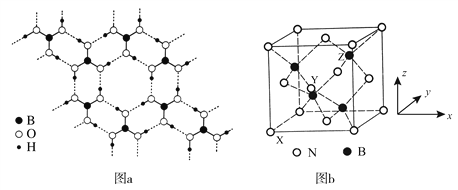
��3�����³�ѹ�����ᣨH3BO3������ṹΪ��״�����άƽ��ṹ��ͼa��
�� Bԭ�ӵ��ӻ���ʽΪ________��������ĽǶȽ�����������ˮ�е��ܽ��С������ʱ�ܽ������________��
�� ·��˹���������Ϊ���κοɽ��ܵ��ӶԵķ��ӻ����ӽ�·��˹�ᣬ�κοɸ������ӶԵķ��ӻ����ӽ�·��˹��ӽṹ�Ƕȷ���������·��˹�________��
��4������������BN�����������ĥ���������ϣ��侧���ṹ����ʯ���ƣ���ͼb��ʾ��
�� �뵪ԭ��ֱ�����ӵ���ԭ�ӹ��ɵļ�����״Ϊ________����ԭ�Ӻ͵�ԭ�������ӵ���С��Ϊ________Ԫ����
�� ��������������Ҫ�أ�
ԭ�������������ʾ�����ڲ���ԭ�ӵ����λ�á���ͼb��ʾ������ԭ���������XΪ��0��0��0����Yԭ�ӵ��������Ϊ��1/2��0��1/2������Zԭ�ӵ��������Ϊ________��
�������������������Ĵ�С����״����֪������������ܶ�Ϊd gcm��3�������ӵ�����ֵΪNA��������a��________nm�����г�����ʽ������
���𰸡� 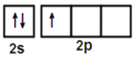 С N C2H6 sp2 ������Ӽ�ͨ������ϣ�����ʱ����������ƻ��� ���������Bԭ����һ��2p�չ���� �������� �� (
С N C2H6 sp2 ������Ӽ�ͨ������ϣ�����ʱ����������ƻ��� ���������Bԭ����һ��2p�չ���� �������� �� (![]() ��
�� ![]() ��
�� ![]() )
) 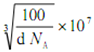
����������1��Bԭ��ԭ�Ӻ���5�����ӣ���̬Bԭ�ӵ����Ų�ʽ�ǣ�1s22s22p1�����Ի�̬Bԭ�ӵļ۵��ӹ������ʽΪ��![]() ��Bԭ�Ӽ۵����Ų�Ϊ2s22p1��Beԭ�Ӽ۵����Ų�Ϊ2s2����Ȼp�ܼ������ϸ���������"���ع���"���ȼ۹��ȫ�����߰�������ȫ��������ȶ��Ľṹ����B��һ�����ܱ�BeС��
��Bԭ�Ӽ۵����Ų�Ϊ2s22p1��Beԭ�Ӽ۵����Ų�Ϊ2s2����Ȼp�ܼ������ϸ���������"���ع���"���ȼ۹��ȫ�����߰�������ȫ��������ȶ��Ľṹ����B��һ�����ܱ�BeС��
��2�������飨NH3BH3���У�N��B�Ļ��ϼ۶���-3��BH3��NH3�Ļ��ϼ۶���0������֮����ͨ����λ�������ĵ���B��������������������������Hԭ������ȱ���������Ӿʹ��ȶ��ṹ����N�������H����һ���µ��Ӷԣ�����N�ṩ�µ��ӶԶ�B�ṩ�չ���γ���λ�����ȵ����������ͬ�ĵ�����Ŀ��ԭ����Ŀ��C2H6�백���黥Ϊ�ȵ�������
��3������ԭ�������ֻ��3�����ӣ�����ԭ���γ�3�Թ��õ��Ӷԣ��۲���Ӷ�Ϊ3���ӻ������Ϊ3���ӻ�����Ϊsp2�����ڵ�H3BO3����֮����Ҫͨ���������������ʱ����������ױ��ƻ���������������ˮ�е��ܽ��С������ʱ�ܽ����������������ĵ�Bԭ��sp2�ӻ�����3��sp2��1��2p����������������4��B��3���۵������ֱ���3��sp2����к����ɼ�����ô�ɼ���Ŀ��3����ĵ��Ӷ��õ���������û�й¶Ե�������ô����һ���չ����Ҳ����2P�չ��������·��˹������ۣ��κοɽ��ܵ��ӶԵķ��ӻ����ӽ�·��˹�ᣬ����������·��˹�ᡣ
��4��������������BN����ͼb��N(��ɫ)ռ���������8�������6�����ģ�������8��![]() +6��
+6��![]() =4����B(��ɫ)ռ��С�����壨�˷�֮һ�������������ģ���4������һ����������4��N��4��B���뵪ԭ��ֱ�����ӵ���ԭ�ӹ��ɵļ�����״Ϊ�������壬���е�ԭ�������ģ�4����ԭ���ڶ��㣻��ԭ�Ӻ͵�ԭ�������ӵ���С��Ϊ��Ԫ������ԭ�������������ʾ�����ڲ���ԭ�ӵ����λ�á���ͼb��ʾ������ԭ���������XΪ��0��0��0����Yԭ�ӵ��������Ϊ��1/2��0��1/2����ZΪB(��ɫ)ռ��С�����壨�˷�֮һ������������������֪Z���ڵ��������
=4����B(��ɫ)ռ��С�����壨�˷�֮һ�������������ģ���4������һ����������4��N��4��B���뵪ԭ��ֱ�����ӵ���ԭ�ӹ��ɵļ�����״Ϊ�������壬���е�ԭ�������ģ�4����ԭ���ڶ��㣻��ԭ�Ӻ͵�ԭ�������ӵ���С��Ϊ��Ԫ������ԭ�������������ʾ�����ڲ���ԭ�ӵ����λ�á���ͼb��ʾ������ԭ���������XΪ��0��0��0����Yԭ�ӵ��������Ϊ��1/2��0��1/2����ZΪB(��ɫ)ռ��С�����壨�˷�֮һ������������������֪Z���ڵ��������![]() ������Zԭ�ӵ��������Ϊ(
������Zԭ�ӵ��������Ϊ(![]() ��
�� ![]() ��
�� ![]() )������֪������������ܶ�Ϊd gcm��3�������ӵ�����ֵΪNA����������Ϊa��N�����ԭ������Ϊ14��B�����ԭ������Ϊ11������ݾ�����������ʽ��d��a3=
)������֪������������ܶ�Ϊd gcm��3�������ӵ�����ֵΪNA����������Ϊa��N�����ԭ������Ϊ14��B�����ԭ������Ϊ11������ݾ�����������ʽ��d��a3=![]() ������a=
������a=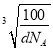 cm=
cm=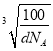 ��107 nm��
��107 nm��

����Ŀ��ʵ�����Ʊ�1��2����������ķ�Ӧԭ�����£�CH3CH2OH ![]() CH2=CH2��H2O��CH2=CH2��Br2
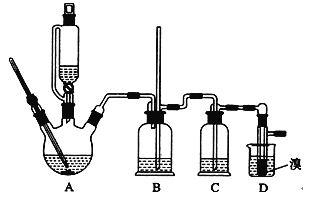
CH2=CH2��H2O��CH2=CH2��Br2![]() BrCH2CH2Br��������������������Ҵ��Ʊ�l��2�����������װ����ͼ��ʾ���й��������±���
BrCH2CH2Br��������������������Ҵ��Ʊ�l��2�����������װ����ͼ��ʾ���й��������±���
�Ҵ� | 1��2���������� | ���� | |
״̬ | ��ɫҺ�� | ��ɫҺ�� | ��ɫҺ�� |
�ܶ�/g��cm-3 | 0.79 | 2.2 | 0.71 |
�е�/�� | 78.5 | 132 | 34.6 |
�۵�/�� | ��130 | 9 | ��116 |
�ش��������⣺
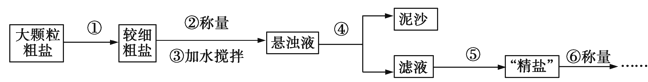
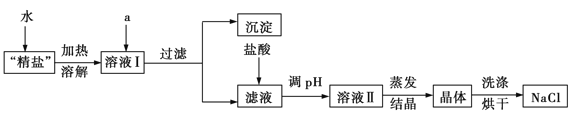
����װ��c��Ӧ����______��ѡ����ţ�����Ŀ�������շ�Ӧ�п������ɵ��������塣
��ˮ ��Ũ���� ������������Һ �ܱ���̼��������Һ
�ƽ���������ֲ�Ʒ���ڷ�Һ©���м�ˮ�����ã�����Ӧ��ˮ��________�㡣����ϡ����¡���
���ж�d�����Ʊ��������鷴Ӧ�ѽ������������_________________________��
����������������δ��Ӧ��Br2�������________������ȷѡ��ǰ����ţ�ϴ�ӳ�ȥ��
��ˮ ������������Һ �۵⻯����Һ ���Ҵ�
����1��2����������Ϊԭ�ϣ��Ʊ�������ϩ��Ϊ�����ԭ�������ʣ���ͬѧ������������̣�1��2����������ͨ�����٣���Ӧ�Ƶã��ڣ�����ͨ�����ۣ���Ӧ�Ƶ�����ϩ��������ϩ�Ƶþ�����ϩ��
��______________���Ӧ���ͣ����仯ѧ����ʽ__________________��
��______________���Ӧ���ͣ���
����Ŀ������ͼ��ʾװ�ý�������ʵ�飺��������Һ������У�Ԥ���������ʵ���������
ѡ�� | �������� | �������� | Ԥ����е����� |
A | Ũ���� | MnO2 | ��������ɫ���� |
B | Ũ���� | ��ɰֽ��ĥ�������� | ��������ɫ���� |
C | �ữ��FeCl2��Һ | H2O2��Һ | ��Һ����ػ�ɫ�������ݲ��� |
D | Fe2(SO4)3��Һ | ͭ�� | ��Һ����ɫ���к�ɫ������� |

A. A B. B C. C D. D
����Ŀ������п�̳�����Ҫ�ɷ�ΪZnO��������CuO��FeO��Ϊԭ�ϣ�������ȡ����п�ͽ���п��
��.��ȡ����п��Ҫ�������£�
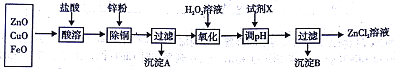
�±��г�����ؽ������������������������pH����ʼ������pH����������Ũ��Ϊl.0mol/L���㣩��
�������� | ��ʼ������pH | ������ȫ��pH |
Fe3+ | 1.1 | 3.2 |
Zn2+ | 5.2 | 6.4 |
Fe2+ | 5.8 | 8.8 |
��1��Ϊ�˼ӿ췴Ӧ������������Ҫ�ʵ����������¶Ȳ���̫�ߣ�ԭ����_________��
��2������H2O2��Һ������Ӧ�����ӷ���ʽ_______��
��3������ͼ����Ϊ�˽�����Һ����ȣ�����pH��ΧΪ_______�����Լ�XΪZn2(OH)2CO3������X�����ʵ����ӷ���ʽΪ________��
��4����֪�������£�Ksp[Fe(OH)3]=4.0��10-38����pH=3ʱ����Һ��c(Fe3+)Ϊ______����ZnCl2��Һ����ȡ��ˮZnCl2�ķ�����__________.
��.��ȡ����п���ü��ܽ�ZnO(s)+2NaOH (aq)+H2O=Na2[Zn(OH)4](aq),Ȼ�����ȡҺ��
��5����п�̳����ü��ܣ������������ܺ�������Ҫԭ����______��
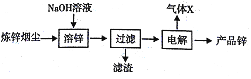
��6����ʯī���缫���ʱ����������������Ϊ______�������ĵ缫��ӦΪ______��