��Ŀ����
����Ŀ��������ԭ��Ӧ��һ����Ҫ�ķ�Ӧ���밴Ҫ�����������Ŀ��
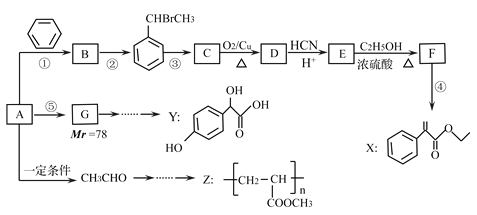
(1)�Է�ӦNH3+O2����NO+H2O(δ��ƽ)�����������õ����ŷ��������ת�Ƶķ������Ŀ�� ______________________________���÷�Ӧ�У�________�ǻ�ԭ����________�ǻ�ԭ�������ԭ��������________��
(2)��һ�������£���Ӧ2NH3+3CuO![]() 3Cu+N2+3H2O��˳�����У��Դ˷�Ӧ�ķ�����������______��
3Cu+N2+3H2O��˳�����У��Դ˷�Ӧ�ķ�����������______��
���÷�Ӧ���û���Ӧ
����Ӧ��NH3������ΪN2
���ڷ�Ӧ�������˽���ͭ�Ļ�ԭ��
���ڷ�Ӧ��ÿ����1 mol H2Oת��1 mol����
(3)�ڷ�Ӧ2H2S+SO2![]() 3S+2H2O�б�������Ԫ���뱻��ԭ��Ԫ�ص�������Ϊ ________��
3S+2H2O�б�������Ԫ���뱻��ԭ��Ԫ�ص�������Ϊ ________��
���𰸡�(1)![]()
(2)��
(3)2��1
��������(1)�ɻ��ϼ۱仯��֪��NH3�ǻ�ԭ����O2����������NO���������������ǻ�ԭ���H2O�ǻ�ԭ���
(2)��Ӧ����û�е��ʲ��룬��һ�������û���Ӧ����������Ӧ��NԪ�ػ��ϼ���3���ߵ�0����NH3������ΪN2������ȷ��NH3�ڷ�Ӧ�����ֻ�ԭ�ԣ��������ɻ��ϼ۱仯��֪���÷�Ӧת��6e����ÿ����1 mol H2Oת��2 mol���ӣ�������
(3)�ڷ�Ӧ��H2S��SԪ�ر�������SO2��SԪ�ر���ԭ������������֮��Ϊ2��1��

 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д�����Ŀ������������ClNO�����л��ϳ��е���Ҫ�Լ�������NO��Cl2��Ӧ�õ�����ѧ����ʽΪ
2NO(g)+Cl2(g) ![]() 2ClNO(g)
2ClNO(g)
��1���о����������������ڴ����к������ӵ������ʱ�������������ȣ��漰���·�Ӧ��
��2NO2(g)+NaCl(s)![]() NaNO3(s)+ClNO(g) ��H1 K1
NaNO3(s)+ClNO(g) ��H1 K1
��4NO2(g)+2NaCl(s)![]() 2NaNO3(s)+2Cl2(g)+2NO(g) ��H2 K2
2NaNO3(s)+2Cl2(g)+2NO(g) ��H2 K2
��2NO(g)+Cl2(g)![]() 2ClNO(g) ��H3 K3
2ClNO(g) ��H3 K3
��H1 ����H2 ����H3 ֮��Ĺ�ϵʽΪ______��ƽ�ⳣ��K1��K2��K3֮��Ĺ�ϵʽΪ_______��
��2����֪���ֻ�ѧ���ļ����������£�
��ѧ�� | NO�еĵ����� | Cl-Cl�� | Cl-N�� | ClNO�е�N=O�� |
����/(KJ/mol) | 630 | 243 | a | 607 |
����H3+2a=_________��
��3��300��ʱ��2ClNO(g)![]() 2NO(g)+Cl2(g)������Ӧ���ʵı���ʽΪv��=k��cn(ClNO)(kΪ���ʳ�����ֻ���¶��й�)��������ϳ���Ũ�ȹ�ϵ���±���ʾ��
2NO(g)+Cl2(g)������Ӧ���ʵı���ʽΪv��=k��cn(ClNO)(kΪ���ʳ�����ֻ���¶��й�)��������ϳ���Ũ�ȹ�ϵ���±���ʾ��
���� | c(ClNO)/(mol/L) | v/(mol��L��s) |
�� | 0.30 | 3.60��10-9 |
�� | 0.60 | 1.44��10-8 |
�� | 0.90 | 3.24��10-8 |
n=___________,k=____________��
��4���������ݻ���Ϊ2L�ĺ����ܱ������зֱ����4mol��2mol ClNO���ڲ�ͬ�¶��·�����Ӧ��2ClNO(g) ![]() 2NO(g)+Cl2(g)�����ƽ��ʱClNO��Ũ�����¶ȱ仯��������ͼ��ʾ��ͼ��ABC���λ�������ϣ���
2NO(g)+Cl2(g)�����ƽ��ʱClNO��Ũ�����¶ȱ仯��������ͼ��ʾ��ͼ��ABC���λ�������ϣ���
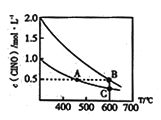
��2ClNO(g) ![]() 2NO(g)+Cl2(g) ��S____0��ѡ����>����<������=������
2NO(g)+Cl2(g) ��S____0��ѡ����>����<������=������
��A��B����ƽ�ⳣ��֮��ΪK(A):K(B)=_____��
��B��C����ClNO��ת����a(B)______a(C) ��ѡ����>����<������=������
��5���ڴ���������NO��COת��Ϊ�����壺
2CO(g)+2NO(g) ![]() 2CO2(g)+N2(g) ��H=-748KJ/mol
2CO2(g)+N2(g) ��H=-748KJ/mol
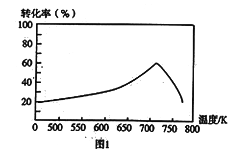
��һ�������£���λʱ���ڲ�ͬ�¶��²ⶨ�ĵ�������ת������ͼ1��ʾ���¶ȸ���710Kʱ�����¶ȵ����ߵ�������ת���ʽ��͵�ԭ�������___________��
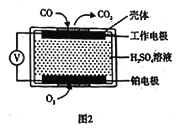
����֪���ⶨ������NO��CO�������õ绯ѧ������������������CO�������Ĺ���ԭ����ͼ2��ʾ�������缫�ķ�ӦʽΪ______��
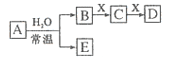
����Ŀ��ijѧϰС�����ʵ�����������ķ�������Ϊ��MnO2��FeCl3��6H2O�����ܲ���Cl2���������������װ����֤�ý��ۡ�
���������ϡ�FeCl3��6H2O���ػ�ɫ���壬�۵�37�棬�е�280��285�档
��ʵ����ơ�

�����۳�����B�з�����Ӧ�Ļ�ѧ����ʽ��___��C�е��Լ���___��
��ʵ����̡�
���� | ���� |
��1������飬װ�����������á� ��2������ҩƷ������װ�á� ��3����ȼ�ƾ��ơ� | i��A�в��ֹ����ۻ����Ϸ����ְ����� ii���Ժ�����ɫ���壬�ܱڸ��Ż�ɫҺ�Σ� iii��B����Һ������ |
��ʵ�������
��1��С������һ����Ϊ����i�еİ���������СҺ�Σ�����������ԭ����___��
��2����������iiʱ�����˷��磬��ͬѧ��Ϊ��FeCl3��6H2O���ػ�ɫ���е㲻�ߣ���ɫ�����غ���FeCl3Ҳ�ᵼ��B����Һ��������Ӧ�����ӷ���ʽ��___��
С��ͬѧͨ������ʵ��֤������ii�Ļ�ɫ�����к���Fe3+��������ʵ�鲹��������
a��ֻ��A�м���FeCl3��6H2O�����ȣ���ʼ�����������Ժ���ֻ�ɫ���壬�ܱ����л�ɫҺ�Ρ�
b��____________________________________ ����Һ��졣
��3��Ϊ��һ��ȷ�ϻ�ɫ���庬�� Cl2��С����A��B ������ʢ�б����Ȼ�����Һ���Թܣ���������ͨ�������Ȼ�����Һ��KI������Һ��KI������Һ������֤ʵ��ɫ�����к���Cl2�������Ȼ�����Һ����Ҫ������___��
��4��A�й����ּ��Ⱥ�õ�Fe2O3��MnCl2�������ʵ���֮��Ϊ1:1����A���ܷ�Ӧ�Ļ�ѧ����ʽ��________���ӻ�����з����Fe2O3�ķ����ǣ�___��
����ȫԤ������ʵ������У����Թ�B����Һ�Ѿ���ʼ����������Ӧ������ȡ��һ����ʩ��___ (��д���)��
a����ȥ�ƾ���
b��ȡ��B��ʢ����Һ���Թ�
c����A��B��ĵ��ܴ��齺����ȡ��