��Ŀ����
����Ŀ���������ȣ�ClO2������ɫ������ˮ�����壩�Ǹ�Ч���Ͷ������������ش��������⣺
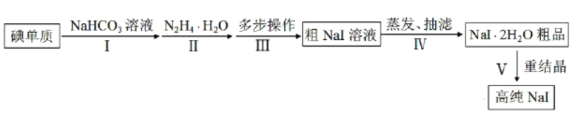
��1��ʵ������NH4Cl�����ᡢNaClO2���������ƣ�Ϊԭ�ϣ�ͨ�����¹����Ʊ�ClO2��
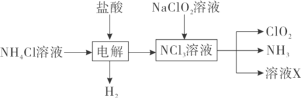
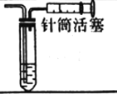
�ٵ��ʱ�����缫��ӦʽΪ__________________________��
�ڳ�ȥClO2�е�NH3��ѡ�õ��Լ���___________�����ţ���
a��ˮ b����ʯ�� c��Ũ���� d������ʳ��ˮ
��2������ͼװ�ÿ��Բⶨ�������ClO2�ĺ�����

������ƿ�м��������ĵ⻯�أ���50mLˮ�ܽ���ټ���3mLϡ���
���ڲ���Һ��װ���м���ˮ��ʹҺ��û������Һ��ܵĹܿڣ�
��һ�����Ļ������ͨ����ƿ�����գ�
����������Һ��װ���е�ˮ������ƿ�У�
������0.1000mol��L-1��������Ʊ���Һ�ζ���ƿ�е���Һ��I2+2S2O32-��2I��+S4O62-����ָʾ����ʾ�յ�ʱ����ȥ20.00mL�����������Һ���ڴ˹����У�
����ƿ��ClO2��⻯�ط�Ӧ�����ӷ���ʽΪ______________________��
�ڲ���Һ��װ�õ�������______________________��
��V�м���ָʾ�����ζ����յ��������______________________��
�ܲ�û������ClO2������Ϊ______g��
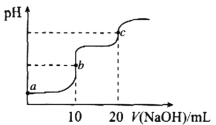
��ijͬѧ��ij���̶ֿ�ģ�������50mL�ζ��ܽ���ʵ�飬���ζ����е�Һ�洦����ͼ��ʾ�Ŀ̶ȴ��������Һ������________������ţ���
a������23.60mL b������27.60mL c����23.60mL d������27.60mL
![]()
���𰸡�NH4+-6e-+3Cl-=NCl3+4H+ c 2ClO2+10I-+8H+=2Cl-+5I2+4H2O ���ղ�����ClO2 ���������һ����������Ʊ���Һ����Һ����ɫ��Ϊ��ɫ���Ұ��������Һ��ɫ���ٸı� 0.027 d
��������
��1�����ʱ���Դ��������������ʧȥ���ӣ�����������Ӧ�����Դ���������������õ����ӷ�����ԭ��Ӧ����������ClO2���Ʊ����̣���֪��������H2��NCl3���ݴ˿ɵó��缫��Ӧ����ʽ��
��2������ʵ����̣���֪ʵ��������漰�ķ�Ӧ����ʽ�У�2ClO2+10I-+8H+=2Cl-+5I2+4H2O��I2+2S2O32-��2I��+S4O62-���ݴ˽��з�����
��1�������ʱ����ʧȥ���ӣ�����������Ӧ��������е��������ɵIJ����֪�����缫��Ӧ����ʽΪ��NH4+-6e-+3Cl-=NCl3+4H+����Ϊ��NH4+-6e-+3Cl-=NCl3+4H+��
������ClO2��NH3�����ʽ��������
a��ClO2��NH3��������ˮ��������ˮ�ͱ���ʳ��ˮ����ȥ������a�����
b����ʯ�Ҳ������հ�����b�����
c��Ũ����������հ������Ҳ�Ӱ��ClO2���ʳ�ȥ��ȥClO2�е�NH3��ѡ�õ��Լ�Ũ���ᡣc����ȷ��
d��ClO2��NH3��������ˮ�������ñ���ʳ��ˮ����ȥ������d�����
����c��
��2�����������Ϣ��֪��ClO2ͨ����ƿ��I-����ΪI2����������ԭΪCl-��ͬʱ����ˮ������ƿ��ClO2��⻯�ط�Ӧ�����ӷ���ʽΪ��2ClO2+10I-+8H+=2Cl-+5I2+4H2O����Ϊ��2ClO2+10I-+8H+=2Cl-+5I2+4H2O��
������ClO2�����壬���ӷ��������У����Բ���Һ��װ�õ����������ղ����ClO2���壬��Ϊ�����ղ�����ClO2��
�����ݵ��������������Һ��Ӧ�Ǽ����˵�����ָʾ����������ζ��յ�ʱ��I2��ȫת��ΪI-����Һ��ɫ��ȥ���ʵζ����յ�������ǣ����������һ����������Ʊ���Һ����Һ����ɫ��Ϊ��ɫ���Ұ��������Һ��ɫ���ٸı䣻��Ϊ�����������һ����������Ʊ���Һ����Һ����ɫ��Ϊ��ɫ���Ұ��������Һ��ɫ���ٸı䣻
�����ݷ�Ӧ����ʽ��2ClO2+10I-+8H+=2Cl-+5I2+4H2O��I2+2S2O32-��2I��+S4O62-���ɵù�ϵʽ��2ClO2��5I2��10S2O32-���ɴ˿�֪���������ClO2������Ϊ![]() ����Ϊ��0.027��
������0.027��
��ͼ��Һ�����Ϊ22.40mL���ζ���������Ϊ50.00mL����Һ�������������Һ�������Ϊ27.60mL����Ϊ�ζ��ܼ��첿������һ����Һ�壬��ζ�����Һ�����Ӧ����27.60mL����ѡd����Ϊ��d��

 100�ִ�����ĩ���ϵ�д�
100�ִ�����ĩ���ϵ�д� ��У���˿��ֿ���ϵ�д�
��У���˿��ֿ���ϵ�д�����Ŀ����������ʵ��װ���������Ӧʵ�����
ѡ�� | װ��ͼ | ʵ��Ŀ�� |
A |
| �ռ������HCl |
B |
| ��ȥCO2�к��е�����HCl |
C |
| �ռ�H2��NH3��CO2��Cl2��HCl��NO��NO2������ |
D |
| ���װ�õ������� |
A.AB.BC.CD.D