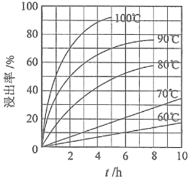
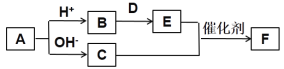
��Ŀ����
����Ŀ��������0.1mol/L��NaHCO3��Һ250mL��������в��裺
��1���õ�����ƽ��ȡNaHCO3����______________g��
��2�����ƺõ�NaHCO3�������______________��������������ˮ����___________��
��3������Һ_____________����Һ����______________����250mL����ƿ�У��ڲ��������в�����ʧ�κ�һ��Һ�壬�����ʹ��Һ��Ũ��ƫ_________����ͣ���
��4������������ˮ��ϴ_______________2-3�Σ���ϴ��Һ����______________�У�
��5��������ƿ�ڼ�ˮ������̶���______________ʱ������________________С�ļ�ˮ��___________________������ˮ�����̶��ߣ��������ҺŨ��ƫ_________����ͣ���Ӧ��_____________��
��6�����Ǻ�ƿ�ǣ�___________��Ȼ����õ���Һ�����Լ�ƿ�У����ñ�ǩ��
���𰸡�2.100 �ձ� �ܽ� ��ȴ������ ������ �� �ձ��ڱںͲ����� ����ƿ 1cm-2cm ��ͷ�ι� ��Һ�İ���ײ����ø��̶������� �� ���� ҡ��
��������
������0.1mol/L��NaHCO3��Һ250mL����Ҫ��NaHCO3�����ʵ���![]() ������
������![]() ,�ʴ�Ϊ��2.100g��
,�ʴ�Ϊ��2.100g��
�ƽ��ƺõ�NaHCO3��������ձ��У�������������ˮ�����ܽ⣬�ʴ�Ϊ���ձ����ܽ⣻
�Ǵ���Һ��ȴ�����º���Һ���Ų���������250mL����ƿ�У��ڲ��������в�����ʧ�κ�һ��Һ�壬��ʧҺ�壬ʵ�ʾ����������ʵ������٣�Ũ��ƫ�ͣ��ʴ�Ϊ����ȴ�����£����������ͣ�
������������ˮ��ϴ���������ձ��ڱ�2-3�Σ���ϴ��Һ��������ƿ�У��ʴ�Ϊ���ձ��ڱںͲ�����������ƿ��
��������ƿ�ڼ�ˮ������̶���1cm-2cmʱ�����ý�ͷ�ι�С�ļ�ˮ����Һ�İ���ײ����ø��̶������У�����ˮ�����̶��ߣ���Һ���ƫ��Ũ��ƫ�ͣ�Ӧ���������ʴ�Ϊ��1cm-2cm����ͷ�ιܣ���Һ�İ���ײ����ø��̶������У��ͣ�������
�����Ǻ�ƿ�ǣ�ҡ�ȣ�Ȼ����õ���Һ�����Լ�ƿ�У����ñ�ǩ���ʴ�Ϊ��ҡ�ȣ�

 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д�����Ŀ��I.��ˮMgBr2������������ʵ���Ҳ���þм��Һ��Ϊԭ���Ʊ���ˮMgBr2��װ����ͼ��ʾ(�г�װ��ʡ��)��
��֪����Mg��Br2��Ӧ���ҷ��ȣ�MgBr2����ǿ��ˮ��
��MgBr2+3C2H5OC2H5![]() MgBr2��3C2H5OC2H5��
MgBr2��3C2H5OC2H5��

��Ҫ�������£�
����1��������ƿ��װ��10gþм��150mL��ˮ���ѣ�װ��C�м���15mLҺ�壻
����2������ͨ�����������ֱ������ȫ����������ƿ�У�
����3����Ӧ��Ϻ�ָ������£����˳�ȥþ����Һת������һ�������ƿ�У���ȴ��0�棬�������壬�ٹ��˵������Ѻ��廯þ��Ʒ��
����4����������CCl4�ܽ��Ʒ����ȴ��0�棬���羧�壬���ˣ�ϴ�ӵ������Ѻ��廯þ��������160��ֽ����ˮMgBr2��Ʒ��
��ش���������
(1)����A��������___________��B������Ϊ___________��
(2)�����йز���4��˵����ȷ����___________(��ѡ����ĸ)
A.����75%���Ҵ�����CCl4�ܽ��Ʒ
B.������160�����ҪĿ���dz�ȥCCl4
C.ϴ�Ӿ����ѡ��0���CCl4
D.�ò���ֻ��ȥ�˿��ܲ�������
(3)����װ��C��Ϊװ��D�����ܻᵼ�µĺ����___________��
(4)Ϊ�ⶨ��Ʒ�Ĵ��ȣ�����EDTA(��дΪY4��)����Һ�ζ����Ը���T(��дΪIn)Ϊָʾ������֪Mg2+�����T��EDTA�����γ�������EDTA��Mg2+��ϸ��ȶ���
���� | ��ɫ | ���� | ��ɫ |
Y4- | ��ɫ | MgY2- | ��ɫ |
In | ����ɫ | MgIn- | �ƺ�ɫ |
��Ӧ�����ӷ���ʽΪMg2++Y4��=MgY2��
�ٵζ��յ������Ϊ______________________��
�ڲⶨǰ���ȳ�ȡ0.2500g��ˮMgBr2��Ʒ���ܽ����2�θ���T��Һ��ָʾ������0.0500mol��L��1��EDTA����Һ�ζ����յ㣬����EDTA����Һ25.00mL��������ˮMgBr2��Ʒ�Ĵ�����______________________(������������ʾ)��
��.������Mg5Al3(OH)19(H2O)4������������ȼ���ϣ�����ʱ�����»�ѧ����ʽ�ֽ⣺2Mg5Al3(OH)19(H2O)4===27H2O+10MgO+3Al2O3��
(5)д���û���������ȼ�����������ݣ�______________________��
(6)MgO������NH4Cl��ˮ��Һ�������ӷ���ʽ��ʾ��ԭ����______________________��