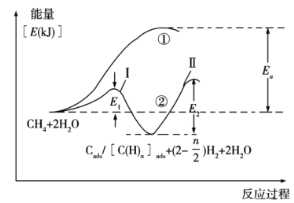
��Ŀ����
����Ŀ��ijС����Fe2+����ʵ���й۲쵽�쳣����Ϊ̽������ɫ��ȥ����ԭ��������ʵ�飺
��� | ʵ��I | ʵ��II | ʵ��III |
ʵ�鲽�� |
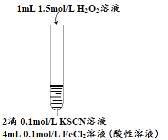
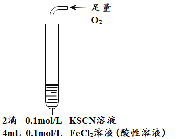
| ��ʵ��I��ɫ�����Һ�����ݷֱ����ʵ��
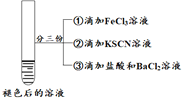
| Ϊ��һ��̽������ɫ��ȥ����ԭ���ֽ�������ʵ�� �� ��ȡ��Ӧ�����Һ���μ������BaCl2��Һ |
���� | ��Һ�ȱ�죬Ƭ�̺��ɫ��ȥ�����������ɣ�������ΪO2�� | ������������ ����Һ��� �۲�����ɫ���� | ����Һ��죬һ��ʱ�����ɫ�� ���ް�ɫ�������� |
������������ʵ�飬������������ȷ����
A.�ڴ�ʵ��������H2O2����Fe2�������ʱ�����SCN�������ʿ�
B.ͨ��ʵ����Ƴ�ʵ����к�ɫ��ȥ��ԭ��������SCN��������
C.ͨ��ʵ����ʵ���Ա��Ƴ���ɫ��ȥֻ��H2O2���������й�
D.����������ʵ����к�ɫ��ȥ��ԭ���뻯ѧƽ���ƶ�ԭ����
���𰸡�D
��������
�Ȼ������ữ����Һ�е���KSCN��Һ������������ⷢ��������ԭ��Ӧ��ͨ��������������Һ����Ҳ���ɫ��˵���������������������������ӣ��μ������BaCl2��Һ���ް�ɫ�����������ᱵ�������ɣ��ų������ɵ���������SCN-�Ŀ��ܣ�
A. �ڴ�ʵ��������H2O2����Fe2�������ʱ�����SCN�������ʿ죬������Һ�ȱ�죬��SCN������ȫ������ɫ��ȥ����A��ȷ��
B. ʵ��II�еμ�FeCl3��Һ����죬˵����SCN-���μ������BaCl2��Һ���а�ɫ�������ɣ�˵��SCN-������ΪSO42-�������Ƴ�ʵ�����к�ɫ��ȥ��ԭ��������SCN������������B��ȷ��
C. �Ա�ʵ���ʵ����ų��������ӷ�Ӧ������SCN-���ӷ�Ӧ��ʵ����ų������ɵ���������SCN-�Ŀ��ܣ����к�ɫ��ȥ�����ж�������������H2O2��������SCN-��������Ϊ��������ӣ���C��ȷ��
D. ��ɫ����ΪFe3+��SCN-����γɵ�����Ϊ���淴Ӧ����SCN-��������ƽ�������ƶ�����ɫ��ȥ���뻯ѧƽ���ƶ�ԭ���йأ���D����
��ѡD��

����Ŀ�����б�Ŵ���Ԫ�����ڱ��е�һ����Ԫ�أ��û�ѧʽ�ش��������⣺
| ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
�� | �� | �� | �� | |||||
�� | �� | �� | �� | �� | �� | |||
�� | �� | �� |
(1)�١��ۡ��ݵ�����������ˮ���������ǿ����˳��Ϊ___________(�ѧʽ����ͬ)��
(2)�ڡ��ۡ����γɵļ������Ӱ뾶�ɴ�С��˳����______________��
(3)��͢������������Ӧ��ˮ���ﻯѧʽΪ_______��_________�����������е�ȼ���ɵ���ɫ�Ĺ��壬�ù���ĵ���ʽΪ_______________�����ܹ���ˮ���ҷ�Ӧ����д���÷�Ӧ�Ļ�ѧ����ʽ_______________����Ԫ�آ�ĵ���ͨ��NaBr��Һ�У���Ӧ�����ӷ���ʽΪ_______________��
(4)�ޡ��ߡ�������Ԫ���γɵ���̬�⻯����ȶ���������____________(�ѧʽ����ͬ)������Ԫ�طǽ�������ǿ������˳��Ϊ___________��
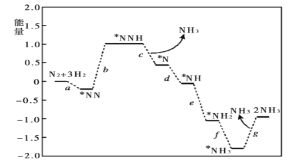
����Ŀ��AlN���Ͳ���Ӧ��ǰ���㷺�����Ʊ��������о���Ϊ�ȵ㡣
����������£�
���� | �۵�/�� | �е�/�� | ��N2��Ӧ�¶�/�� | ��Ӧ������ֽ��¶�/�� |
Al | 660 | 2467 | ��800 | AlN����2000 ����1400������ AlCl3������181������ |
Mg | 649 | 1090 | ��300 | Mg3N2����800 |
(1)AlN���Ʊ���
�� ��ѧ�����������
��.һ���¶��£���AlCl3�����NH3Ϊԭ���Ʊ�AlN����Ӧ�Ļ�ѧ����ʽ��____________________��
��.������Ӧ���˵��¶ȷ�Χ��______�棨����ĸ����
a.75~100 b.600~1100 c.2000~2300
�� ����ֱ�ӵ�������
Al��N2��ֱ�ӻ���ΪAlN���壬AlN�ܽ�Al��������Ӧ���Լ������С������¶ȣ���Al���о��Ȳ�������Mg�ۣ���ʹAl����ȫ��ת��ΪAlN���塣�ù��̷����ķ�Ӧ�У�__________________��_________��2Al + N2 ![]() 2AlN��
2AlN��
��̼�Ȼ�ԭ����
��Al2O3��C��ʯī����N2Ϊԭ�ϣ��ڸ������Ʊ�AlN��
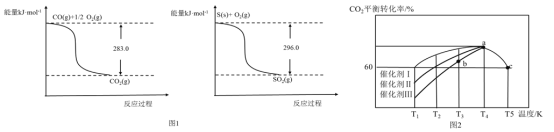
��֪����. 2Al2O3(s) 4Al(g) + 3O2(g) H 1 =��3351 kJ��mol-1
��. 2C(ʯī��s) + O2(g) = 2CO(g) H 2 =��221 kJ��mol-1
��. 2Al(g) + N2(g) = 2AlN(s) H 3 =��318 kJ��mol-1
����ƽ���ƶ�ԭ��������Ӧ���Է�Ӧ���Ŀ���Ӱ�죺______________________________________��
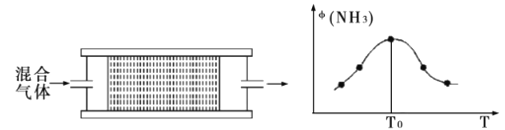
(2)AlN�����ʡ�AlN��ĩ�ɷ���ˮ�⡣��ͬ�����£���ͬ������AlN��ĩˮ��ʱ��ҺpH�ı仯��ͼ��ʾ��
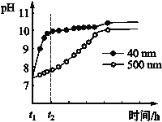
�� AlN��ĩˮ��Ļ�ѧ����ʽ��____________________________________��
�� ����t1-t2ʱ�����������߲���Ŀ���ԭ��_______________________________��
(3)AlN������⡣��a g AlN��Ʒ�м�������ŨNaOH��Һ��Ȼ��ͨ��ˮ������NH3ȫ����������NH3�ù�����v1 mL c1 mol��L-1 H2SO4��Һ������ȫ��ʣ���H2SO4��v2 mL c2 mol��L-1 NaOH��Һǡ���кͣ�����Ʒ��AlN������������________________________________��