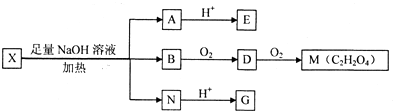
��Ŀ����
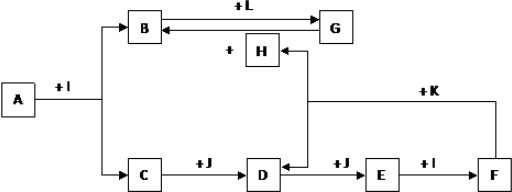
������AΪһ�ֳ�������Ԫ����һ�ֳ����ǽ���Ԫ����ɵĻ�����������������ӵĸ�����Ϊ2��3�� KΪ������̬�ǽ������ʣ�JΪ������̬���ʣ�����Ϊ���������I��F�ڳ�����ΪҺ̬��C��DΪ�̼������壬H��ɫ��ζ���壬BΪ��ɫ��״������LΪ�ȼҵ�еij�����Ʒ��F��Ũ��Һ��K���ȿ�����D��H��������������δ�����
��1��H�ĽṹʽΪ�� ��B�Ļ�ѧʽΪ��
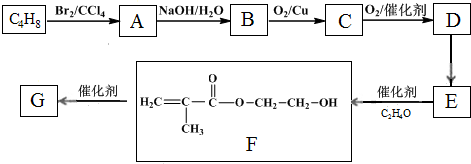
��2��д�����б仯�Ļ�ѧ����ʽ��A+I��B+C�� F��Ũ��Һ��K���ȿ�����D��H��
��3��д�����б仯�����ӷ���ʽ��G��ˮ��Һ��ͨ�˹�����H��
��4����A��K��������������������ʵ���_ �����ڷǵ���ʵ��� ���ñ�Żش𣩣�

��1��H�ĽṹʽΪ��
��2��д�����б仯�Ļ�ѧ����ʽ��A+I��B+C��
��3��д�����б仯�����ӷ���ʽ��G��ˮ��Һ��ͨ�˹�����H��
��4����A��K��������������������ʵ���_
���㣺������ƶ�
ר�⣺�ƶ���
������F��Ũ��Һ�볣����̬�ǽ�������K���ȿ����ɴ̼�������D����ɫ��ζ����H����ѧ����ϸ÷�Ӧ��ֻ��̼��Ũ�������Ũ���ᷴӦ����KΪ̼��C��J������Ӧ�õ�E��E��Һ��I��Ӧ�õ�F������֪JΪO2��IΪH2O�����ӻ�����A��ˮ�з���ˮ�ⷴӦ�õ�B��C��BΪ��ɫ��״������LΪ�ȼҵ�еij�����Ʒ�����߷�Ӧ�õ�G������֪BΪAl��OH��3��LΪNaOH��GΪNaAlO2����FΪŨ�������ͼ�е����ʼ�ת����CΪH2S��DΪSO2��EΪSO3��HΪCO2��AΪһ��Al��S��ɵĻ���������������������ӵĸ�����Ϊ2��3�����ɵ�֪AΪAl2S3���ݴ˽��
���
�⣺F��Ũ��Һ�볣����̬�ǽ�������K���ȿ����ɴ̼�������D����ɫ��ζ����H����ѧ����ϸ÷�Ӧ��ֻ��̼��Ũ�������Ũ���ᷴӦ����KΪ̼��C��J������Ӧ�õ�E��E��Һ��I��Ӧ�õ�F������֪JΪO2��IΪH2O�����ӻ�����A��ˮ�з���ˮ�ⷴӦ�õ�B��C��BΪ��ɫ��״������LΪ�ȼҵ�еij�����Ʒ�����߷�Ӧ�õ�G������֪BΪAl��OH��3��LΪNaOH��GΪNaAlO2����FΪŨ�������ͼ�е����ʼ�ת����CΪH2S��DΪSO2��EΪSO3��HΪCO2��AΪһ��Al��S��ɵĻ���������������������ӵĸ�����Ϊ2��3�����ɵ�֪AΪAl2S3��
��1��HΪCO2���ṹʽΪ��O=C=O��B�Ļ�ѧʽΪ��Al��OH��3���ʴ�Ϊ��O=C=O��Al��OH��3��
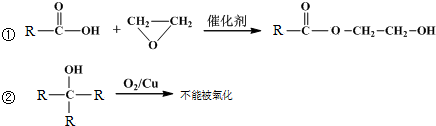
��2��A+I��B+C�Ļ�ѧ����ʽΪ��Al2S3+6H2O=2Al��OH��3+3H2S����
F��Ũ��Һ��K���ȿ�����D��H����Ӧ��ѧ����ʽΪ��C+2H2SO4��Ũ��
CO2��+2SO2��+2H2O��
�ʴ�Ϊ��Al2S3+6H2O=2Al��OH��3+3H2S����C+2H2SO4��Ũ��
CO2��+2SO2��+2H2O��
��3��ƫ�����Ƶ�ˮ��Һ��ͨ������Ķ�����̼����Ӧ������������������̼�����ƣ��÷�Ӧ�����ӷ���ʽΪ��AlO2-+CO2+H2O=Al��OH��3��+HCO3-��
�ʴ�Ϊ��AlO2-+CO2+H2O=Al��OH��3��+HCO3-��
��4��Al2S3��Al��OH��3��H2S�����ᡢH2O��NaOH��NaAlO2���ڵ���ʣ�����Al��OH��3��H2S��H2O����������ʣ�SO2��SO3��CO2���ڷǵ���ʣ�
�ʴ�Ϊ��B��C��I��D��E��H��
��1��HΪCO2���ṹʽΪ��O=C=O��B�Ļ�ѧʽΪ��Al��OH��3���ʴ�Ϊ��O=C=O��Al��OH��3��
��2��A+I��B+C�Ļ�ѧ����ʽΪ��Al2S3+6H2O=2Al��OH��3+3H2S����
F��Ũ��Һ��K���ȿ�����D��H����Ӧ��ѧ����ʽΪ��C+2H2SO4��Ũ��
| ||
�ʴ�Ϊ��Al2S3+6H2O=2Al��OH��3+3H2S����C+2H2SO4��Ũ��
| ||
��3��ƫ�����Ƶ�ˮ��Һ��ͨ������Ķ�����̼����Ӧ������������������̼�����ƣ��÷�Ӧ�����ӷ���ʽΪ��AlO2-+CO2+H2O=Al��OH��3��+HCO3-��
�ʴ�Ϊ��AlO2-+CO2+H2O=Al��OH��3��+HCO3-��
��4��Al2S3��Al��OH��3��H2S�����ᡢH2O��NaOH��NaAlO2���ڵ���ʣ�����Al��OH��3��H2S��H2O����������ʣ�SO2��SO3��CO2���ڷǵ���ʣ�
�ʴ�Ϊ��B��C��I��D��E��H��
���������⿼�������ƶϣ�����ͻ�ƿ��ǣ�F��Ũ��Һ�볣����̬�ǽ�������K���ȿ����ɴ̼�������D����ɫ��ζ����H���ٽ����Ŀ��Ϣ��ת����ϵ�ƶϣ���Ҫѧ����������Ԫ�ػ��������ʣ��ѶȽϴ�

��ϰ��ϵ�д�
 ��ɢ˼ά�¿���ϵ�д�
��ɢ˼ά�¿���ϵ�д�
�����Ŀ
�����£����и���Һ����������ȷ���ǣ�������
| A����֪����HF��CH3COOH��pH��ȵ�NaF��CH3COOK��Һ�У�c��Na+��-c��F-����c��K+��-c��CH3COO-�� |
| B��0.1mol/L�Ĵ����pH=a��0.01mol/L�Ĵ����pH=b����a+1��b |
| C����ͬ�����£�pH=5�Ģ�NH4Cl��Һ����CH3COOH��Һ����ϡ����������Һ����ˮ�������c��H+�����٣��ڣ��� |
| D����ˮ�루NH4��2SO4��Һ��Ϻ�pH=7����Һ�У�c��NH4+����C��SO42-��=1��1 |
ij���������۾�����Ϊ��ͼ��ʾ�ľۺ�������й�˵��������ǣ�������
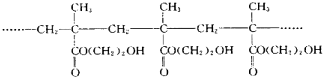

| A�����ɸþۺ���ķ�Ӧ���ڼӾ۷�Ӧ |
| B���þۺ���ĵ����DZ������� |
| C���þۺ�������д��ڴ�����-OH�����Ծ��нϺõ���ˮ�� |
D���þۺ���ĽṹͲʽΪ��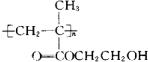 |
���������У��������Ӽ����ǣ�������
| A��Cl2 |
| B��CO2 |
| C��NaCl |
| D��H2O2 |
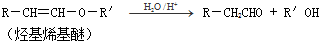
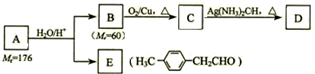
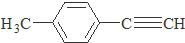 ����һ��·�����£�
����һ��·�����£�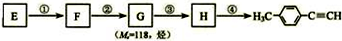
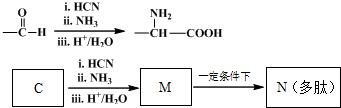
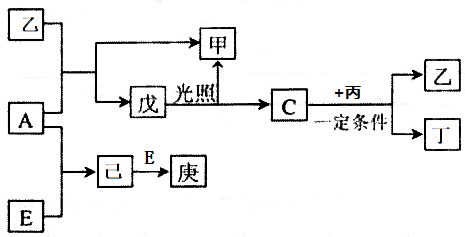
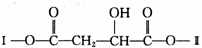 ������I����Ϊδ֪���ֵĽṹ��Ϊ�Ʋ�X�ķ��ӽṹ��������ͼ��ת����
������I����Ϊδ֪���ֵĽṹ��Ϊ�Ʋ�X�ķ��ӽṹ��������ͼ��ת����