��Ŀ����
A��B��C��D��E��ԭ������������������ֶ�����Ԫ�ء�Aԭ�ӵĵ��Ӳ��������ĺ������������ͬ��A��C��B��D�ֱ�ͬ���壬B��C������������֮����E��������������ͬ��A��E�γɵĻ����ﻯѧʽΪAE��������к�18�����ӡ���ش��������⣺
(1)AԪ�ص��ʵĻ�ѧʽΪ________��DԪ�ص�ԭ�ӽṹʾ��ͼΪ_______��
(2)��A��B��C����Ԫ���γɵĻ�����ĵ���ʽΪ________�����ֻ������γɵľ����������Ļ�ѧ��������________��
(3)A��B��D����Ԫ���γɵ�һ�ֻ������A��B�γɵ�һ�ֻ������ܷ���������ԭ��Ӧ����Ӧ�Ļ�ѧ����ʽΪ__________________��
(4)B��C��E����Ԫ���γ�һ���Σ�����������ԭ�ӵĸ�����Ϊ1��1��1����25��ʱ������������ˮ�������ҺpH>7��������________(�����ӷ���ʽ��ʾ)��
(1)H2��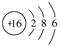 ��(2)
��(2)  �����Ӽ������ۼ���(3)H2SO3��H2O2=H2SO4��H2O��(4)ClO����H2O
�����Ӽ������ۼ���(3)H2SO3��H2O2=H2SO4��H2O��(4)ClO����H2O HClO��OH��
HClO��OH��
����

��ϰ��ϵ�д�
�����Ŀ
ԭ������С��36������Ԫ��X��Y��Z��W���й����ǵ���Ϣ���±���ʾ��
| Ԫ�� | ���ֽṹ֪ʶ | �������� |
| X | Xԭ�Ӻ�������Ų��������ܼ��ϣ�������������������ͬ | X�ж����������XO��XO2��X2O4�ȣ�ͨ�������XO2��X2O4���� |
| Y | Yԭ�ӵ��������������ڴ�����������2�� | Y���γɻ�������������Ԫ�� |
| Z | Zԭ�ӵ���������������4 | ZԪ�ص���������ϼ���������ϼ۴����͵���6 |
| W | W��һ�ֺ���������Ϊ52��������Ϊ28 | ��6��W�Ļ�������ǿ�����ԣ�������Ƥ��ӹ������������˾���Ƿ�Ƽݵ� |
����д���пհ�(����ʱX��Y��Z��W����Ӧ��Ԫ�ط���)��
(1)X���ʷ��ӵĵ���ʽ��________���Ҽ��ͦм��ĸ�������________��
(2)ZԪ��ԭ������㹲��________�ֲ�ͬ�˶�״̬�ĵ��ӡ�Z���⻯��Һ̬ʱ________(��ܡ����ܡ�)���硣
(3)W�Ļ�̬ԭ�Ӻ�������Ų�ʽ��_____________________________��
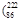 Rn),�Ӷ�����������˺�����ش�:
Rn),�Ӷ�����������˺�����ش�:
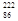 Rn,�ʳ�
Rn,�ʳ�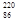 Rn,�ʽ�
Rn,�ʽ�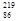 Rn,�ʽ�
Rn,�ʽ�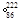 Rn
Rn Rn
Rn Rn����������(����ĸ���)��
Rn����������(����ĸ���)��