ĢāÄæÄŚČŻ
”¾ĢāÄæ”æĮņĖįŹĒ¼«ĘäÖŲŅŖµÄ»Æ¹¤ŌĮĻ£¬ŌŚ¹¤Ņµ”¢Å©Ņµ”¢Ņ½Ņ©”¢¾üŹĀµČĮģÓņÓ¦ÓĆ¹ć·ŗ”£¹¤ŅµÉĻĶس£ÓĆ½Ó“„·ØÖĘĮņĖį£¬Ö÷ŅŖŌĮĻŹĒĮņĢśæóŗĶæÕĘų”£½Ó“„·ØÖĘĮņĖįµÄÉś²ś¹ż³Ģ“óÖĀæÉ·ÖĪŖČżøö½×¶Ī£ŗ¶žŃõ»ÆĮņµÄÖĘČ”ŗĶ¾»»Æ£»¶žŃõ»ÆĮņ×Ŗ»ÆĪŖČżŃõ»ÆĮņ£»ČżŃõ»ÆĮņµÄĪüŹÕŗĶĮņĖįµÄÉś³É”£ĪŖĮĖ·ĄÖ¹»·¾³ĪŪČ¾²¢¶ŌĪ²Ęų½ųŠŠ×ŪŗĻĄūÓĆ£¬ĮņĖį³§³£ÓĆ°±Ė®ĪüŹÕĪ²ĘųµÄSO2”¢SO3µČĘųĢ壬ŌŁĻņĪüŹÕŅŗÖŠ¼ÓČėÅØĮņĖį£¬ŅŌÖĘČ”øßÅØ¶ČµÄSO2¼°£ØNH4£©2SO4ŗĶNH4HSO4¹ĢĢ唣ĪŖĮĖ²ā¶ØÉĻŹö£ØNH4£©2SO4ŗĶNH4HSO4¹ĢĢå»ģŗĻĪļµÄ×é³É£¬ĻÖ³ĘČ”øĆѳʷĖÄ·Ż£¬·Ö±š¼ÓČėĻąĶ¬ÅØ¶ČµÄNaOHČÜŅŗ50.00mL£¬¼ÓČČÖĮ120”ę×óÓŅ£¬Ź¹°±ĘųČ«²æŅŻ³ö[£ØNH4£©2SO4ŗĶNH4HSO4µÄ·Ö½āĪĀ¶Č¾łøßÓŚ200”ę]£¬²āµĆÓŠ¹ŲŹµŃ鏿¾ŻČēĻĀ£Ø±ź×¼×“æö£©£ŗ
ŹµŃé | ѳʷµÄÖŹĮæ/g | NaOHČÜŅŗµÄĢå»ż/mL | °±ĘųµÄĢå»ż/L£Ø±ź×¼×“æö£© |
1 | 7.24 | 50.00 | 1.792 |
2 | 14.48 | 50.00 | 3.584 |
3 | 21.72 | 50.00 | 4.032 |
4 | 36.20 | 50.00 | 2.240 |
£Ø1£©ÓÉ1×鏿¾ŻÖ±½ÓĶĘ²ā£ŗ1.81gѳʷ½ųŠŠĶ¬ŃłŹµŃ鏱£¬Éś³É°±ĘųµÄĢå»ż£Ø±ź×¼×“æö£©ĪŖ___L”£
£Ø2£©ŹŌ¼ĘĖćøĆ»ģŗĻĪļÖŠ£ØNH4£©2SO4ŗĶ NH4HSO4µÄĪļÖŹµÄĮæÖ®±ČĪŖ___”£
£Ø3£©ĒóĖłÓĆNaOHČÜŅŗµÄĪļÖŹµÄĮæÅضČ___mol/L”£
”¾“š°ø”æ0.448 1”Ć2 6
”¾½āĪö”æ
£ØNH4£©2SO4ŗĶ NH4HSO4µÄ»ģŗĻŅŗÖŠ¼ÓČėĒāŃõ»ÆÄĘ£¬ŅĄ“Ī·¢ÉśµÄ·“Ó¦ŹĒ2NH4HSO4+2NaOH=£ØNH4£©2SO4+Na2SO4+2H2O”¢£ØNH4£©2SO4+2NaOH= Na2SO4+2H2O+2NH3”£
£Ø1£©øł¾ŻŹµŃé1”¢2æÉÖŖ£¬Ōö¼ÓѳʷµÄÖŹĮ棬·Å³öµÄ°±ĘųŌö¶ą£¬ĖµĆ÷ŹµŃé1ÖŠĒāŃõ»ÆÄĘ¹żĮ棬7.24gѳʷ֊µÄļ§øłĄė×ÓČ«²æÉś³É°±Ęų·Å³ö£¬Čō¼ÓČė1.81gѳʷ£¬ļ§øłĄė×ÓŅ²ÄÜČ«²æÉś³É°±Ęų·Å³ö£¬Éč·Å³ö°±ĘųµÄĢå»żŹĒVL£¬Ōņ![]() £¬V=0.448L”£
£¬V=0.448L”£
£Ø2£©ŹµŃé1ÖŠ£¬7.24gѳʷ֊µÄļ§øłĄė×ÓČ«²æÉś³É°±Ęų·Å³ö£¬Éčѳʷ֊µÄ£ØNH4£©2SO4ŗĶ NH4HSO4ĪļÖŹµÄĮæ·Ö±šŹĒxmol”¢ymol£¬øł¾ŻµŖŌŖĖŲŹŲŗć£¬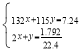 £¬½āµĆx=0.02mol”¢y=0.04mol£¬
£¬½āµĆx=0.02mol”¢y=0.04mol£¬![]() £»
£»
£Ø2£©ÓÉŹµŃé3”¢4æÉŅŌ擳ö£¬ŹµŃé3ŌŁŌö“óѳʷµÄÖŹĮæ£¬Éś³É°±ĘųµÄĢå»ż¼õŠ”£¬ĖµĆ÷ĒāŃõ»ÆÄĘĪļÖŹµÄĮæ²»×ć£¬Éč36.2gѳʷ֊NH4HSO4ĪļÖŹµÄĮæĪŖamol£¬Ōņ![]() £¬a=0.2mol£¬ĻČ·¢Éś·“Ó¦H++OH-=H2O£¬ŌņøĆ·“Ó¦ĻūŗÄĒāŃõ»ÆÄʵÄĪļÖŹµÄĮæĪŖ0.2mol£¬ŌŁ·¢ÉśNH4++OH-
£¬a=0.2mol£¬ĻČ·¢Éś·“Ó¦H++OH-=H2O£¬ŌņøĆ·“Ó¦ĻūŗÄĒāŃõ»ÆÄʵÄĪļÖŹµÄĮæĪŖ0.2mol£¬ŌŁ·¢ÉśNH4++OH-![]() £¬øł¾Ż°±ĘųµÄĢå»żĪŖ2.24L£¬øĆ·“Ó¦ĻūŗÄĒāŃõ»ÆÄʵÄĪļÖŹµÄĮæĪŖ0.1mol£¬ĖłŅŌ50mLĒāŃõ»ÆÄĘČÜŅŗŗ¬ÓŠĒāŃõ»ÆÄʵÄĪļÖŹµÄĮæŹĒ0.3mol£¬ĒāŃõ»ÆÄĘČÜŅŗµÄÅØ¶ČŹĒ
£¬øł¾Ż°±ĘųµÄĢå»żĪŖ2.24L£¬øĆ·“Ó¦ĻūŗÄĒāŃõ»ÆÄʵÄĪļÖŹµÄĮæĪŖ0.1mol£¬ĖłŅŌ50mLĒāŃõ»ÆÄĘČÜŅŗŗ¬ÓŠĒāŃõ»ÆÄʵÄĪļÖŹµÄĮæŹĒ0.3mol£¬ĒāŃõ»ÆÄĘČÜŅŗµÄÅØ¶ČŹĒ![]() 6mol/L”£
6mol/Lӣ

 ŠĀĢāŠĶČ«³Ģ¼ģ²āĘŚÄ©³å“Ģ100·ÖĻµĮŠ“š°ø
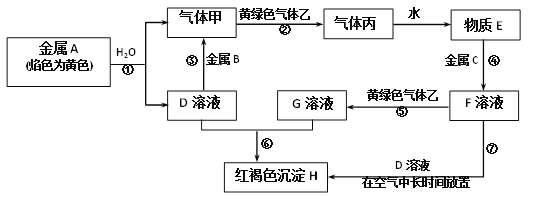
ŠĀĢāŠĶČ«³Ģ¼ģ²āĘŚÄ©³å“Ģ100·ÖĻµĮŠ“š°ø”¾ĢāÄæ”æH2O2ŹĒŹµŃéŹŅ³£¼ūµÄĒæŃõ»Æ¼Į£¬ŌŚŅ½ĮĘÉĻæÉÓĆ×÷Ļū¶¾¼ĮµČ”£
£Ø1£©Ņ»ÖÖÕżŌŚæŖ·¢µÄĄūÓĆO2ŗĶH2O×÷ŌĮĻĶعż»ÆŗĻÖĘČ”H2O2µÄ·½·Ø£¬ĘäŌĄķČēĶ¼ĖłŹ¾”£øĆ·½·ØÖĘČ”H2O2µÄ×Ü»Æѧ·“Ó¦·½³ĢŹ½ĪŖ____”£

£Ø2£©ĪŖĢ½¾æĶā½ēĢõ¼ž¶ŌH2O2·Ö½ā»Æѧ·“Ó¦ĖŁĀŹµÄÓ°Ļģ£¬Ļą¹ŲŹµŃéÉč¼ĘČēĻĀ±ķĖłŹ¾£ŗ
ŹŌ¹Ü ±ąŗÅ | ŹµŃéÄæµÄ | H2O2ČÜŅŗ | ĪĀ¶Č | Ė®µÄ Ģå»ż/mL | FeCl3ČÜŅŗĢå»ż/mL | |
ÖŹĮæ ·ÖŹż | Ģå»ż/mL | |||||
¢ń | ĪŖ±ąŗÅ¢ņŹµŃé²ĪÕÕ | 12% | 5£®0 | ³£ĪĀ | 0 | 0 |
¢ņ | ĪĀ¶Č¶Ō·“Ó¦ĖŁĀŹµÄÓ°Ļģ | £Ø £© | 5£®0 | 60”ę | 0 | 0 |
¢ó | ĪŖ±ąŗÅ¢ōŹµŃé²ĪÕÕ | 4£®0% | 5£®0 | ³£ĪĀ | £Ø £© | 0 |
¢ō | £Ø £© | 4£®0% | 5£®0 | ³£ĪĀ | 0 | 1£®0 |
ĢīŠ“±ķ֊ȱɣµÄÄŚČŻ£ŗ¢ņ_______£»¢ó__________£»¢ō_________”£
£Ø3£©Óɲ¬£ØPtĪ»ÓŚ×ó±ß£©ŗĶ½š£ØAuĪ»ÓŚÓŅ±ß£©×é³ÉµÄÄÉĆ×°ō·ÅČėH2O2ČÜŅŗÖŠ£ØČēĶ¼£©£¬ÄÉĆ×°ō½«·¢Éś¶ØĻņŅĘ¶Æ”£

Ōņ£ŗAuŅ»²ąĪŖµē³ŲµÄ____¼«£ØŃ”Ģī£ŗ”°Õż”±»ņ”°øŗ”±£©£»ÄÉĆ×°ōĻņ____£ØŃ”Ģī£ŗ”°×ó”±»ņ”°ÓŅ”±£©ŅĘ¶Æ”£
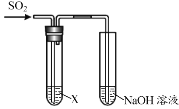
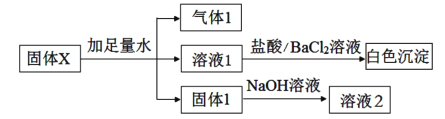
”¾ĢāÄæ”æÄ³Ń§Ļ°Š”×éÓĆĻĀĶ¼×°ÖĆŃŠ¾æSO2µÄŠŌÖŹ”£
| ŠņŗÅ | X | ŹµŃéĻÖĻó |
¢ń | ×ĻÉ«ŹÆČļČÜŅŗ | ĻÖĻóa | |
¢ņ | Ę·ŗģČÜŅŗ | ČÜŅŗÓÉŗģÉ«±äĪŖĪŽÉ«£¬¼ÓČČŗóÓÖ»Öø“ŌĄ“µÄŃÕÉ« | |
¢ó | ĖįŠŌKMnO4ČÜŅŗ | ČÜŅŗÓÉ×ĻÉ«±äĪŖĪŽÉ« |
Ēė»Ų“š£ŗ
£Ø1£©ŹµŃé¢ńÖŠ£¬ĻÖĻóaŹĒ______”£
£Ø2£©øł¾ŻŹµŃé¢ņ£¬ĶʶĻSO2µÄ»ÆѧŠŌÖŹŹĒ______”£
£Ø3£©øł¾ŻŹµŃé¢ó£¬ĶʶĻĪŽÉ«ČÜŅŗÖŠĖłŗ¬µÄĄė×ÓŹĒK+”¢Mn2+”¢H+ŗĶ______”£
£Ø4£©½įŗĻĄė×Ó·½³ĢŹ½ĖµĆ÷ŹµŃéÖŠNaOHČÜŅŗµÄ×÷ÓĆŹĒ______”£
”¾ĢāÄæ”æĮņĖįŹĒ¼«ĘäÖŲŅŖµÄ»Æ¹¤ŌĮĻ£¬ŌŚ¹¤Ņµ”¢Å©Ņµ”¢Ņ½Ņ©”¢¾üŹĀµČĮģÓņÓ¦ÓĆ¹ć·ŗ”£¹¤ŅµÉĻĶس£ÓĆ½Ó“„·ØÖĘĮņĖį£¬Ö÷ŅŖŌĮĻŹĒĮņĢśæóŗĶæÕĘų”£½Ó“„·ØÖĘĮņĖįµÄÉś²ś¹ż³Ģ“óÖĀæÉ·ÖĪŖČżøö½×¶Ī£ŗ¶žŃõ»ÆĮņµÄÖĘČ”ŗĶ¾»»Æ£»¶žŃõ»ÆĮņ×Ŗ»ÆĪŖČżŃõ»ÆĮņ£»ČżŃõ»ÆĮņµÄĪüŹÕŗĶĮņĖįµÄÉś³É”£ĪŖĮĖ·ĄÖ¹»·¾³ĪŪČ¾²¢¶ŌĪ²Ęų½ųŠŠ×ŪŗĻĄūÓĆ£¬ĮņĖį³§³£ÓĆ°±Ė®ĪüŹÕĪ²ĘųµÄSO2”¢SO3µČĘųĢ壬ŌŁĻņĪüŹÕŅŗÖŠ¼ÓČėÅØĮņĖį£¬ŅŌÖĘČ”øßÅØ¶ČµÄSO2¼°£ØNH4£©2SO4ŗĶNH4HSO4¹ĢĢ唣ĪŖĮĖ²ā¶ØÉĻŹö£ØNH4£©2SO4ŗĶNH4HSO4¹ĢĢå»ģŗĻĪļµÄ×é³É£¬ĻÖ³ĘČ”øĆѳʷĖÄ·Ż£¬·Ö±š¼ÓČėĻąĶ¬ÅØ¶ČµÄNaOHČÜŅŗ50.00mL£¬¼ÓČČÖĮ120”ę×óÓŅ£¬Ź¹°±ĘųČ«²æŅŻ³ö[£ØNH4£©2SO4ŗĶNH4HSO4µÄ·Ö½āĪĀ¶Č¾łøßÓŚ200”ę]£¬²āµĆÓŠ¹ŲŹµŃ鏿¾ŻČēĻĀ£Ø±ź×¼×“æö£©£ŗ
ŹµŃé | ѳʷµÄÖŹĮæ/g | NaOHČÜŅŗµÄĢå»ż/mL | °±ĘųµÄĢå»ż/L£Ø±ź×¼×“æö£© |
1 | 7.24 | 50.00 | 1.792 |
2 | 14.48 | 50.00 | 3.584 |
3 | 21.72 | 50.00 | 4.032 |
4 | 36.20 | 50.00 | 2.240 |
£Ø1£©ÓÉ1×鏿¾ŻÖ±½ÓĶĘ²ā£ŗ1.81gѳʷ½ųŠŠĶ¬ŃłŹµŃ鏱£¬Éś³É°±ĘųµÄĢå»ż£Ø±ź×¼×“æö£©ĪŖ___L”£
£Ø2£©ŹŌ¼ĘĖćøĆ»ģŗĻĪļÖŠ£ØNH4£©2SO4ŗĶ NH4HSO4µÄĪļÖŹµÄĮæÖ®±ČĪŖ___”£
£Ø3£©ĒóĖłÓĆNaOHČÜŅŗµÄĪļÖŹµÄĮæÅضČ___mol/L”£