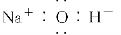��Ŀ����
�̲��и�����Na2O2��H2O��Ӧ�Ļ�ѧ����ʽ2Na2O2��2H2O=4NaOH��O2����Ϊ��̽��Na2O2��H2O��Ӧ�Ļ�����ijѧϰ̽��С���ڽ�ʦָ�����������ͼ��ʾװ�ý���ʵ�顣

ʵ�鲽�����£�
����ͼʾ��װ�����������װ��������Ϊ���ú�װ��ҩƷ��
������K1��K2�رգ���ע�����е�����ˮ�����Թ��У���ʱ�Թ��в������������
����ѹװ�з�̪�Ľ�ͷ�ιܣ�ʹ��̪�����Թ��У��Թ�����Һ�Ժ�ɫ��
����ѹװ��ϡ����Ľ�ͷ�ιܣ�ʹϡ��������Թ��У���ɫ��ʧ���ٵμ�2�Ρ�
����˫����������A���Թ��й�����ʹ�Թ�����Һͨ����ͷ����B��֧���У����ֵ��ۣ�KI��Һ������KMnO4��Һ��ɫ��
��Ѹ�ٴ�K2���ر�K1��������A���Թ��й��������Թ�����Һ����C���Թ���Լ����֮һʱֹͣ������Ȼ������ˮԡ����C���Թ�Ƭ�̣�������ð����������Ϊ������
��ش��������⣺
��1�������ӷ���ʽ��ʾ���ۣ�KI��Һ������ԭ��_____________________________
________________________________________________________________________��
��2�������ӷ���ʽ��ʾKMnO4��Һ��ɫ��ԭ�� ______________________________
________________________________________________________________________��
��3��A���ñ�����ԡ��C������ˮԡ�����÷ֱ���________��______________________��
��4��Na2O2��H2O��Ӧ�Ļ�����____________________���û�ѧ����ʽ��ʾ����
��1��2I����2H����H2O2=I2��2H2O
��2��2MnO4-��5H2O2��6H��=2Mn2����5O2����8H2O
��3����ֹ���ɵ�H2O2�ֽ⡡ʹH2O2�ֽ�
��4��Na2O2��2H2O=2NaOH��H2O2,2H2O2 2H2O��O2��
2H2O��O2��
����������ʼ�Թ������������ɣ������̪����Һ���ɫ��˵��Na2O2��H2O��Ӧ�����˼��H2O2��Ȼ������ϡ�����к�NaOH��H2O2��C�зֽ�����O2����1��H2O2��ǿ������������I������2��H2O2ʹKMnO4��Һ��ɫ���������仹ԭ�ԡ���3��A���ñ�����ԡ����ֹ�¶ȹ���H2O2�ֽ⣬C������ˮԡ��ʹH2O2�ֽ⡣��4��Na2O2��H2O��Ӧ����H2O2��H2O2���ȷֽ⡣

��¯������ұ��������Ҫ��������������Ҫ��ӦΪ��Fe2O3��s����3CO��g��??2Fe��s����3CO2��g��
��H��a kJ��mol��1��
��1����֪����Fe2O3��s����3C��s��ʯī��=2Fe��s����3CO��g��
��H1����489.0 kJ��mol��1��
��C��s��ʯī����CO2��g��=2CO��g������H2����172.5 kJ��mol��1����a��________��
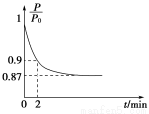
��2��ұ������Ӧ��ƽ�ⳣ������ʽK��________���¶����ߺ�Kֵ________����������������������������С������
��3����T ��ʱ���÷�Ӧ��ƽ�ⳣ��K��64����2 L�����ܱ����������У��ֱ��±���ʾ�������ʣ���Ӧ����һ��ʱ���ﵽƽ�⡣
| Fe2O3 | CO | Fe | CO2 |
��/mol | 1.0 | 1.0 | 1.0 | 1.0 |
��/mol | 1.0 | 2.0 | 1.0 | 1.0 |
����������CO��ƽ��ת����Ϊ________��
������˵����ȷ����________������ĸ����
a���������������ܶȺ㶨ʱ����־��Ӧ�ﵽƽ��״̬
b������Fe2O3�������������CO��ת����
c����������CO��ƽ��ת���ʴ����ҵ�ƽ��ת����
d���ס��������У�CO��ƽ��Ũ��֮��Ϊ2��3
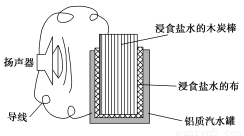
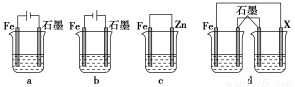
��4����ȡһ����ʩ�ɷ�ֹ������ʴ������װ���е��ձ����ʢ�е�Ũ�ȡ��������NaCl��Һ��
����a��b��cװ�����ܱ���������________������ĸ����
������dװ�ñ�������X���ĵ缫����Ӧ��________�������ƣ���
��5��25 ��ʱ�й����ʵ��ܶȻ����£�Ksp[Mg��OH��2]��5.61��10��12��Ksp[Fe��OH��3]��2.64��10��39��25 ��ʱ������Mg2����Fe3������Һ�еμ�NaOH��Һ�������ֳ�����������Һ��pH��8ʱ��c��Mg2������c��Fe3������________��