��Ŀ����
����Ŀ��I. ��һ�������£����淴ӦA��B![]() mC�仯��ͼ��ʾ����֪�������ʾ�ڲ�ͬ�¶Ⱥ�ѹǿ��������C�ڻ�����е�����������pΪ��Ӧ��T2�¶�ʱ�ﵽƽ�����������ѹ�ı仯�����
mC�仯��ͼ��ʾ����֪�������ʾ�ڲ�ͬ�¶Ⱥ�ѹǿ��������C�ڻ�����е�����������pΪ��Ӧ��T2�¶�ʱ�ﵽƽ�����������ѹ�ı仯�����
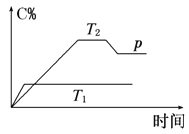
��1���¶�T1________T2(����ڡ������ڡ���С�ڡ�)��
��2������Ӧ��________��Ӧ(����ȡ����ȡ�)��
��3�����A��B��C��Ϊ���壬��m________2(����ڡ������ڡ���С�ڡ�)��
��4�����¶Ⱥ��ݻ�����ʱ������ƽ����ϵ�м���һ������ijϡ�����壬����ϵ��ѹǿ________(�������С�����䡱)��ƽ��_________________�ƶ�(�������Ӧ�������淴Ӧ������)��
II. CO�������ʻ���ѧƷ�Ļ���ԭ�ϣ� 850��ʱ���ں����ܱ�������ͨ��CO��H2O(g)��������CO(g)��H2O(g) ![]() H2(g)��CO2(g)
H2(g)��CO2(g) ![]() H��0���ⶨŨ����ʱ���ϵ���±���
H��0���ⶨŨ����ʱ���ϵ���±���
t/min | c(CO)/ mol��L��1 | c(H2O)/ mol��L��1 |
0 | 0.30 | 0.20 |
2 | \ | 0.10 |
3 | 0.18 | \ |
4 | \ | 0.08 |
�ش���������
��1��t=3minʱ����(��)____________��(��)��ѡ���������������������������
��2��0��2min��CO��ƽ����Ӧ����Ϊ ____________��
��3��������Ӧ�е�COƽ��ת����Ϊ____________��
��4����һ�������£��������з�Ӧ��CO(g)��H2O(g) ![]() H2(g)��CO2(g)�����淴Ӧ��������ʱ��仯������ͼ��ʾ����t1ʱ�ı��ij�ַ�Ӧ����������_____������ţ���
H2(g)��CO2(g)�����淴Ӧ��������ʱ��仯������ͼ��ʾ����t1ʱ�ı��ij�ַ�Ӧ����������_____������ţ���

a������COŨ��
b����ѹ
c������ʱ�������
d��ʹ�ô���
���𰸡����� ���� ���� ���� �� �� ![]() 40% bd
40% bd
��������
I.��1)�¶�Խ�߷�Ӧ����Խ�죬����ƽ���ʱ��Խ�̣�
(2)��ͼ��֪�¶�T1��T2���¶�Խ��C%ԽС���������¶�ƽ�����淴Ӧ�ƶ���
(3)��ͼ��֪��T2�¶�ʱ�ﵽƽ�����������ѹ��C%��С��������ѹǿƽ�����淴Ӧ�ƶ���
(4)���¶Ⱥ��ݻ�����ʱ����ƽ����ϵ�м���һ������ijϡ�����壬��ϵѹǿ������Ӧ������Ũ�Ȳ��䣬ƽ�ⲻ�ƶ���
II.(1)(2)(3)��������ʽ���м��㣬���Եó�3minĩc(H2O)��˵����ʱ�Ѿ��ﵽ��ѧƽ�⣬���û�ѧ��Ӧ���ʵļ��㹫ʽ�������CO�ķ�Ӧ���ʺ�![]() �����������CO��ת���ʣ����(4)���ͼʾ��Ϣ����������ԭ�����н��
�����������CO��ת���ʣ����(4)���ͼʾ��Ϣ����������ԭ�����н��
I.(1)��ͼ��֪���¶�ΪT1�ȵ���ƽ�⣬�¶�Խ�߷�Ӧ����Խ�죬����ƽ���ʱ��Խ�̣���T1��T2���ʴ�Ϊ�����ڣ�
(2)��ͼ��֪�¶�T1��T2���¶�Խ��C%ԽС���������¶�ƽ�����淴Ӧ�ƶ���������ӦΪ���ȷ�Ӧ���ʴ�Ϊ�����ȣ�
(3)��ͼ��֪��T2�¶�ʱ�ﵽƽ�����������ѹ��C%��С��������ѹǿƽ�����淴Ӧ�ƶ�������ѹǿƽ�����������ʵ�����С�ķ����ƶ���˵���÷�Ӧ�淴Ӧ�������������ʵ�����С�ķ���m��2����Ϊ�����ڣ�
(4)���¶Ⱥ��ݻ�����ʱ����ƽ����ϵ�м���һ������ijϡ�����壬����������ʵ���������ϵѹǿ������Ӧ������Ũ�Ȳ��䣬�����淴Ӧ���ʲ��䣬ƽ�ⲻ�ƶ����ʴ�Ϊ��������
II. (1)��������ʽ���㣬�ܹ��ܿ�����3minĩ��c(H2O)=0.08 mol��L��1��4minĩһ����˵��t=3minʱ��Ӧ�Ѿ��ﵽ��ѧƽ�⣬��(��) = ��(��)���ʴ�Ϊ������
(2)���ݷ�Ӧ����ʽ��֪��0��2min CO�����ʵ���Ũ�ȱ仯��0.1 mol��L��1����ƽ����Ӧ����Ϊ![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��
(3)�ӱ������ݿ�֪���ﵽƽ��ʱ��CO��ƽ��Ũ��Ϊ0.18 mol��L��1����������Ӧ�е�COƽ��ת����Ϊ��![]() ���ʴ�Ϊ��40%
���ʴ�Ϊ��40%
(4)��ͼ�п�֪���ı��������淴Ӧ����ͻȻ�����������ƽ�ⲻ�ƶ���
a������COŨ�ȣ�����Ӧ����ͻȻ�����淴Ӧ����������������䣬������ͼʾ��Ϣ��a�������⣻
b������CO(g)��H2O(g)![]() H2(g)��CO2(g)��Ӧǰ����������ʵ������ֲ��䣬�ʼ�ѹʹ�������淴Ӧ����ͬ�ȷ��ȵ�ͻȻ����ƽ�ⲻ�ƶ�������ͼʾ��Ϣ��b�������⣻
H2(g)��CO2(g)��Ӧǰ����������ʵ������ֲ��䣬�ʼ�ѹʹ�������淴Ӧ����ͬ�ȷ��ȵ�ͻȻ����ƽ�ⲻ�ƶ�������ͼʾ��Ϣ��b�������⣻
c������ʱ�����������ϵ��ѹǿ�����Ƿ�Ӧ�����������Ũ�Ⱦ�δ�ı䣬�������淴Ӧ���ʾ����䣬������ͼʾ��Ϣ��c�������⣻
d��ʹ�ô����ܹ�ͬ�ȷ��ȵ�ͻȻ���������淴Ӧ���ʣ�ƽ�ⲻ�ƶ�������ͼʾ��Ϣ��d�������⣻
�ʴ�Ϊ��bd��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�