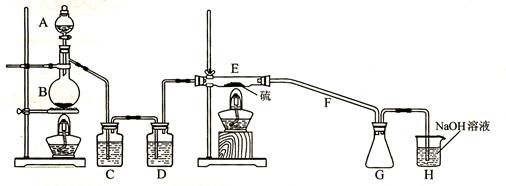
��Ŀ����
��12�֣������Ĺ������ƣ�CaO2��������ˮ���Ҵ��������½�Ϊ�ȶ���CaO2��8H2O��0��ʱ�ȶ���������130��ʱ��Ϊ��ˮCaO2����ʵ���ҿ��ø�����ȡCaO2��8H2O���پ���ˮ�Ƶ�CaO2�����Ʊ��������£�
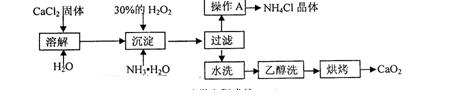
����������Ϣ��ͬ���������⣺
��1��������������ȡCaO2��8H2O�Ļ�ѧ����ʽ�� ��
��2������A�IJ���Ϊ ��
��3�����顰ˮϴ���Ѿ�ϴ���ķ����� ��
��4���ⶨ��Ʒ��CaO2�ĺ�����ʵ�鲽���ǣ���֪��I2+2S2O32-=2I-+S4O62-����
��һ����ȷ��ȡa g��Ʒ����ƿ�У�������������ˮ������b g KIƷ�壬�ٵ�������2 mol��L-1��H2SO4��Һ����ַ�Ӧ��
�ڶ�������������ƿ�м��뼸�ε�����Һ��
����������μ���Ũ��Ϊc mol��L-1��Na2S2O3��Һ����Ӧ��ȫ������Na2S2O3��ҺVmL��
�ٵ�������Ӧ��ȫʱ������Ϊ ��
�ڲ�Ʒ��CaO2����������Ϊ ������ĸ��ʾ����
��ijͬѧ��ʵ���ò�Ʒ��CaO2����������ƫ�ߣ����ƫ�ߵ�ԭ���ǣ��ⶨ�������ɲ��������������Բ��ƣ������ӷ���ʽ��ʾ�� ��

����������Ϣ��ͬ���������⣺
��1��������������ȡCaO2��8H2O�Ļ�ѧ����ʽ�� ��
��2������A�IJ���Ϊ ��
��3�����顰ˮϴ���Ѿ�ϴ���ķ����� ��
��4���ⶨ��Ʒ��CaO2�ĺ�����ʵ�鲽���ǣ���֪��I2+2S2O32-=2I-+S4O62-����
��һ����ȷ��ȡa g��Ʒ����ƿ�У�������������ˮ������b g KIƷ�壬�ٵ�������2 mol��L-1��H2SO4��Һ����ַ�Ӧ��
�ڶ�������������ƿ�м��뼸�ε�����Һ��
����������μ���Ũ��Ϊc mol��L-1��Na2S2O3��Һ����Ӧ��ȫ������Na2S2O3��ҺVmL��
�ٵ�������Ӧ��ȫʱ������Ϊ ��
�ڲ�Ʒ��CaO2����������Ϊ ������ĸ��ʾ����
��ijͬѧ��ʵ���ò�Ʒ��CaO2����������ƫ�ߣ����ƫ�ߵ�ԭ���ǣ��ⶨ�������ɲ��������������Բ��ƣ������ӷ���ʽ��ʾ�� ��
��1��CaCl2+H2O2+2NH3��H2O+6H2O=CaO2��8H2O��+2NH4Cl
��2������Һ�м�������HCl������Ũ������ȴ�ᾧ�����ˣ�ϴ�ӣ�����
��3��ȡ����ϴ��Һ�������м���ϡ�����ữ����������Һ������������ɫ��������˵���Ѿ�ϴ��
��4������ Һ��ɫ��ȥ��
Һ��ɫ��ȥ��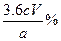 ����4H++4I-+O2=2I2+2H2O��
����4H++4I-+O2=2I2+2H2O��
��2������Һ�м�������HCl������Ũ������ȴ�ᾧ�����ˣ�ϴ�ӣ�����
��3��ȡ����ϴ��Һ�������м���ϡ�����ữ����������Һ������������ɫ��������˵���Ѿ�ϴ��
��4������
 Һ��ɫ��ȥ��
Һ��ɫ��ȥ��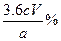 ����4H++4I-+O2=2I2+2H2O��
����4H++4I-+O2=2I2+2H2O����

��ϰ��ϵ�д�
�����Ŀ
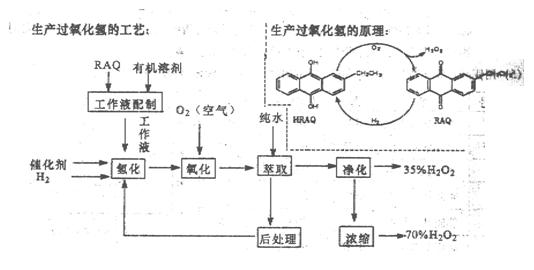

 2SCl2
2SCl2