��Ŀ����
��ѧ��һ����ʵ��Ϊ�����Ŀ�ѧ��
��1��������������ȷ����______������ţ���
| A����Һ©�����ζ��ܺ�����ƿʹ��ǰ�������Ƿ�©ˮ |
| B������ˮ�����Һ©�����ټ������Ҵ�����������ã��ɴӵ�ˮ����ȡ�� |
| C���ྻ��������ʳ��ˮ�н���һ��ʱ�䣬�����������ݣ�˵�������������ⸯʴ |
| D����˿�������о���ȼ�գ��������䣬���ɺ�ɫ���� |
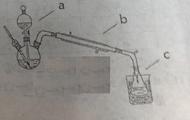
��2����ͭƬ��ϡ���ᷴӦ��ȡNO���壬��ͼװ�����ʺϵ���______������ţ���װ��B�е��Լ������______����װ�õ�������______________________��

��1��AE��ȫ�Ե�2�֣�ֻѡһ������ȷ��1�֣���ѡ���ѡ��0�֣�
��2��ͼ�ң�1�֣� ˮ��1�֣�����ȥNO2���ռ�NO��2�֣�ÿ��Ҫ��1�֣�
�����������:��1��A����Һ©�����ζ��ܺ�����ƿʹ��ǰ�������Ƿ�©ˮ��A��ȷ��B���Ҵ���ˮ���ܣ�������Ϊ��ȡ��ˮ�е����ȡ�������������Ȼ�̼�ȣ�B����ȷ��C��ʳ��ˮ�����ԣ�������������������ʴ��C����ȷ��D����˿�������о���ȼ�գ��������䣬�����غ�ɫ�����Ȼ�����D����ȷ��E������к͵ζ�ʵ���У���ƿ������ˮϴ����ֱ��ʹ�ã������ñ�Һ��ϴ�����ζ���������ˮϴ��������ϴ��ʹ�ã�E��ȷ����ѡAE��
��2������NO���ױ�������������NO2�����Ա���������ˮ���ռ�NO�����Bװ����ʢ�ŵ�ˮ������ΪNO2��ˮ��Ӧ��ͬʱҲ���ܵ���Һ�嵹����������õ�װ����ͼ�ҡ�
���㣺���鳣����ѧʵ�����������NO���Ʊ����ռ��Լ����ӵ�

������ҹ�������������ŷŵķ�����ʯ����ȡ���Ტ����ˮ��ļ����о���óɹ�����������������ͼ��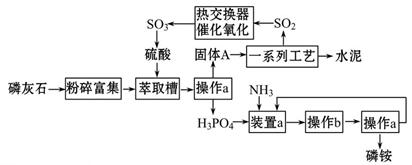
�ش��������⣺
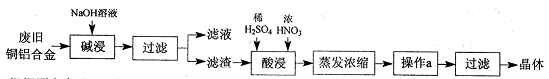
��1������a��������___________��ʵ�����н��д˲����ķDz�����������Ʒ��________________����ʵ�����в���b��������______________________��
��2��װ��a������������ʽ�Σ����ǵĻ�ѧʽ�ֱ���_______________��
��3��������²����A��һ�����е����ʵĻ�ѧʽ��____________________(�ᾧˮ���ֲ�д)��
��4���Ƚ�������ʵ�����Ƚ�����װ�á���ѧʵ����Ҳ���������Ƚ�����ʵ��ij��ʵ��Ŀ�ģ�����Һ�Ƚ���ʱͨ��ʹ�õ�������________________________��
��5����������������β�����˺���N2��O2�⣬������SO2������SO3�������������ڲⶨ����β����SO2��������___________________��
| A��NaOH��Һ����̪��Һ | B��KMnO4��Һ��ϡ���� |
| C����ˮ��������Һ | D����ˮ����̪��Һ |
̼��������ҹ���Ҫ�ĵ���Ʒ��֮һ���������������������ӷ���ʧ��Ϊ�˼�����������ȷ�����ʩ����������ⶨ�京������
��ijѧ�������һ���Բⶨ������̼������Ӳⶨ�������ķ���������Ʒ����Բ����ƿ�У�

��1����ѡ���Ҫ��װ�ã���������������˳��Ϊ ��
��2����Һ©���е�Һ�����ʺϵ��� ��
| A��ϡ���� | B��ϡ���� | C��Ũ���� | D���������� |
����������гɷ��ǣ�NH4��2SO4��������ü�ȩ���ⶨ����������ȩ���ǻ��ڼ�ȩ��һ������������ã������൱�����ᣬ��ӦΪ2��NH4��2SO4+6HCHO����CH2��6N4 +2H2SO4 + 6H2O,���ɵ��������������Ʊ���Һ�ζ����Ӷ��ⶨ���ĺ������������£�
��1���ò�������ȡ���壨NH4��2SO4��Ʒ0��6g���ձ��У�����Լ30mL����ˮ�ܽ⣬�������100mL��Һ���� �����ʽ����ʽ�����ζ���ȷȡ��20��00mL����Һ����ƿ�У�����18%���Լ�ȩ��Һ5mL������5min����1~2�� ָʾ������֪�ζ��յ��pHԼΪ8��8������Ũ��Ϊ0��08mol/L�������Ʊ���Һ�ζ����������±���
| �ζ����� | �ζ�ǰ������mL�� | �ζ��������mL�� |
| 1 | 1��20 | 16��21 |
| 2 | 3��00 | 18��90 |
| 3 | 4��50 | 19��49 |
��ζ��յ�ʱ������Ϊ ���ɴ˿ɼ��������Ʒ�еĵ�����������Ϊ ��
��2���ڵζ�ʵ��������ֵζ��õļ�ʽ�ζ��ܲ��������ڳ��������ݣ��ζ���ʼʱ�����ݣ����ʵ��ⶨ�ĺ�������ʵ��ֵ ���ƫ��ƫС������Ӱ�족����
������ⶨ̼������еĺ�����ʱ��ʹ�ü�ȩ���Ƿ���� ����ǡ����������� ��
��ˮ��Ӧ��ǰ�������Ļ���ԭ����Դ,�Ӻ�ˮ�п���ȡ���ֻ���ԭ��.��ͼ�ǹ�ҵ�϶Ժ�ˮ�ļ����ۺ����õ�ʾ��ͼ����֪����XΪ��ⱥ��ʳ��ˮ���ã�ĸҺ��±����Ҫ����Ca2+��Mg2+,Cl-,SO42-��Br-������)��ش�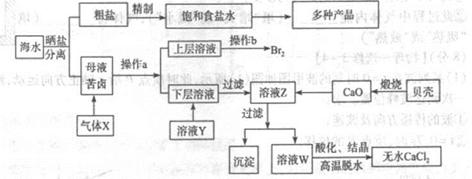
��1���ڴ����к���Ca2+��Mg2+��SO42-�����ʣ�����ʱ���õ��Լ�Ϊ:
| A������ | B���Ȼ�����Һ | C������������Һ | D��̼������Һ��������Լ���˳����(����)______ |
��3��������ҺY��Ŀ����______����CaO������ҺZ��pH,���Գ�ȥMg2+�õ���ҺW���ɱ������ݿ�֪�������Ͽ�ѡ��pH���Χ��______���ữ��ҺWʱ��ʹ�õ��Լ�Ϊ______

��4��������X����ͨ������װ������֤����X�����ʣ�
��ͨ������X��A�г��ֵ�������____________
��Cװ���з�����Ӧ�����ӷ���ʽΪ____________��
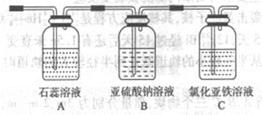
��������С��ͬѧ���һ��ʵ�飬֤��ϴ��ƿB�е�Na2SO3�ѱ�����(����ʵ�鲽�裩
________________________________________________________________________
 N2+3Cu+3H2O�з�Ӧ��CuO��������H2O���������Բⶨͭ�Ľ������ԭ��������ʵ��װ�ã����ȼ��г�װ��δ���������¡�ʵ�鿪ʼʱ��Ӧ�ȵ�ȼ �����A������B�������ƾ��ƣ�c�м�ʯ�ҵ�����Ϊ ��
N2+3Cu+3H2O�з�Ӧ��CuO��������H2O���������Բⶨͭ�Ľ������ԭ��������ʵ��װ�ã����ȼ��г�װ��δ���������¡�ʵ�鿪ʼʱ��Ӧ�ȵ�ȼ �����A������B�������ƾ��ƣ�c�м�ʯ�ҵ�����Ϊ ��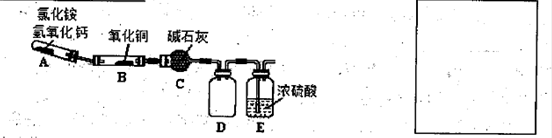
 ��Cl���ȣ�����ȡ�ϴ������Ȼ��ؾ��弰Һ�壬�������£�
��Cl���ȣ�����ȡ�ϴ������Ȼ��ؾ��弰Һ�壬�������£�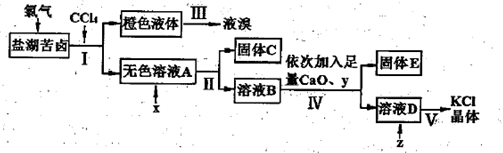

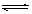 CH3COOC2H5+H20
CH3COOC2H5+H20