��Ŀ����
ʵ������Ҫ0��2mol/L CuSO4��Һ250ml��ʵ���ҿ��ṩ������Һ���Լ��У�����ɫ��������(CuSO4��5H2O) ��4mol/L CuSO4��Һ
(1)���۲��ú����Լ��������ƣ�ʵ������õ��IJ����������ձ�������������ͷ�ι��⣬���ٻ���Ҫ��һ��������________����ʹ�ø�����ǰ������еIJ����� ��
(2)���õ�������������ƣ�����������ƽ��ȡCuSO4�� 5H2O������Ϊ________g�������4mol/L��CuSO4��Һϡ�����ƣ�������Ͳ��ȡ___________ml4mol/L CuSO4��Һ��
(3)ʵ������4mol/L������ͭ��Һϡ��������Һ�����ʵ�鲽���У�
������ȷ�IJ���˳��Ϊ
�����ձ��м���Լ100mlˮ���г���ϡ�ͣ���ȴ������
������Ͳ��ȡһ�����4mol/L ������ͭ��Һ��һ�ձ���
�ۼ�������4mol/L ����ͭ��Һ�����
�ܽ���Һ�ߵ�ҡ�Ⱥ�ת�����Լ�ƿ
�ݼ�ˮ��Һ��������ƿ1-2cm�����ý�ͷ�ιܽ��ж���
��ϴ���ձ��Ͳ�����2-3�β���ϴ��Һע������ƿ������ҡ������ƿ��ʹ��Һ��Ͼ���
�߽���Һת��������ƿ
(4)������Һ�����У������������������Խ���к�Ӱ�죨�ƫ�ߡ�����ƫ�͡�����Ӱ�족��
�������ݺ�����ƿ������ҡ�ȣ����ú�Һ�治���̶��ߣ��ټ�ˮ���̶���
B������ʱ���ӿ̶���
C������ƿδ���T����������Һ
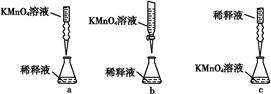
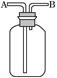
��5��ʵ��������õ�����ˮ����ͼΪʵ������ȡ����ˮ��װ��ʾ��ͼ��ͼ�е��������ԵĴ����� ______________________________��_______________________________��ʵ��ʱA�г�������������ˮ�⣬�����������___ ____���������Ƿ�ֹ���С�
��1��250mL����ƿ��1�֣��� ����Ƿ�©Һ��1�֣���
��2��12��5��1�֣��� 12��5��1�֣���
��3��???????��2�֣���
��4��ƫ�ͣ�1�֣��� ƫ�ߣ�1�֣��� ��Ӱ�죨1�֣���
��5���¶ȼƵ�ˮ����Ӧ��������ƿ֧�ܿڴ���1�֣��� ������ӦΪ�¿ڽ�ˮ���Ͽڳ�ˮ����1�֣��� ���Ƭ����ʯ����1�֣���
�������������
��1��ʵ������Ҫ0��2mol/L CuSO4��Һ250ml��������Ҫ250mL����ƿ��
ʹ�ø�����ǰ������еIJ���Ҫ����Ƿ�©Һ������Ӱ��ʵ�����ݡ�
CuSO4�����ʵ���n=cV=0��25L��0��2mol?L-1=0��05mol��CuSO4?5H2O�����ʵ�������CuSO4�����ʵ���������CuSO4?5H2O������0��05mol��250g/mol=12��5g��
�����4mol/L��CuSO4��Һϡ�����ƣ�������Ͳ��ȡ4mol/L CuSO4��Һ�����Ϊ��0��05mol/(4mol/L )=0��0125L=12��5mL��
��3��???????
��4��A�����ݺ�����ƿ������ҡ�ȣ����ú�Һ�治���̶��ߣ�������Һ������ƿ����ƿ��֮�䣬�ټ�ˮ���̶��ߣ�������Һ�����ƫ����ҺŨ��ƫС��
B������ʱ���ӿ̶��ߣ�����������Һ�����ƫ����ҺŨ��ƫС��
C����Һ�������ˮ���ݣ�����ƿδ���T����������Һ����������ҺŨ����Ӱ�죻
��5���¶ȼƵ�ˮ����Ӧ��������ƿ֧�ܿڴ���������ӦΪ�¿ڽ�ˮ���Ͽڳ�ˮ�� ���Ƭ����ʯ����
���㣺һ�����ʵ���Ũ�ȵ���Һ������

K3[Fe��C2O4��3]��3H2O[���������������ؾ���]������ˮ���������Ҵ�������Ϊ�л���Ӧ�Ĵ�����ʵ���ҿ�����мΪԭ���Ʊ�����ط�Ӧ�������¡���ش��������⣺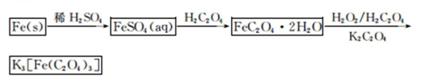
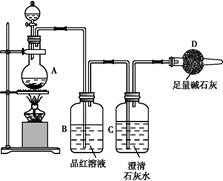
��1����м�г�����Ԫ�أ�������Ʊ�FeSO4ʱ������ж���H2S���壬�������������������Һ���ա���������װ����ȷ���� ������ţ���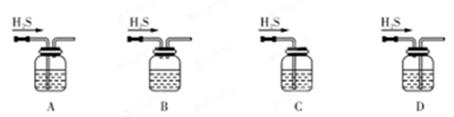
��2���ڵõ���FeSO4��Һ�������������H2 SO4�ữ��Ŀ���� ���õ�K3[Fe(C2O4)3]��Һ�����Ҵ���Ŀ���� ��
��3�������������ᾧˮ��ͨ�������������ⶨ����Ҫ�����У��ٳ����������ں������ѽᾧˮ������ȴ���ܳ��������ظ��ڡ��������أ����㡣
����ݵ�Ŀ���� ��
��4��C2O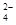 �ɱ�����KMnO4��Һ����ΪCO2���壬��ʵ�������K3[Fe��C2O3��3]��3H2O�����ⶨ����KMnO4����Һ�ζ���
�ɱ�����KMnO4��Һ����ΪCO2���壬��ʵ�������K3[Fe��C2O3��3]��3H2O�����ⶨ����KMnO4����Һ�ζ���
��д���ζ������з�����Ӧ�����ӷ���ʽ ��
�����еζ�������ʹ�ζ����ƫ�ߵ��� ������ţ���
| A���ζ���������ˮϴ�Ӻ�����װ���Һ |
| B����ƿ��װ����Һǰδ�ô���Һ��ϴ |
| C���ζ�ǰ�ζ��ܼ��촦�����ݣ��ζ���������ʧ |
| D����ȡ��Һ���ʱ���ζ�ǰ���Ӷ������ζ����Ӷ��� |
��ѧ��һ����ʵ��Ϊ�����Ŀ�ѧ��
��1��������������ȷ����______������ţ���
| A����Һ©�����ζ��ܺ�����ƿʹ��ǰ�������Ƿ�©ˮ |
| B������ˮ�����Һ©�����ټ������Ҵ�����������ã��ɴӵ�ˮ����ȡ�� |
| C���ྻ��������ʳ��ˮ�н���һ��ʱ�䣬�����������ݣ�˵�������������ⸯʴ |
| D����˿�������о���ȼ�գ��������䣬���ɺ�ɫ���� |
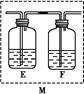
��2����ͭƬ��ϡ���ᷴӦ��ȡNO���壬��ͼװ�����ʺϵ���______������ţ���װ��B�е��Լ������______����װ�õ�������______________________��

ʵ���ҳ����ü�ȩ��HCHO�����ⶨ(NH4)2SO4��Ʒ�е��������������䷴Ӧԭ��Ϊ��4NH4�� ��6HCHO =3H����6H2O��(CH2)6N4H�� �۵ζ�ʱ��1 mol (CH2)6N4H���� l mol H���൱��Ȼ����NaOH����Һ�ζ���Ӧ���ɵ��ᡣij��ȤС���ü�ȩ������������ʵ�飺
����I ��ȡ��Ʒ1��500 g��
����II ����Ʒ�ܽ����ȫת�Ƶ�250 mL����ƿ�У����ݣ����ҡ�ȡ�
����III ��ȡ25��00 mL��Ʒ��Һ��250 mL��ƿ�У�����10 mL 20�������Լ�ȩ��Һ��ҡ�ȡ�����5 min����1~2�η�̪��Һ����NaOH����Һ�ζ����յ㡣�����������������ظ�2�Ρ�
��1�����ݲ���III��գ�
�ټ�ʽ�ζ���������ˮϴ�Ӻ�ֱ�Ӽ���NaOH����Һ���еζ���������Ʒ�е�����������________(�ƫ�ߡ�����ƫ�͡�����Ӱ�족)��
����ƿ������ˮϴ�Ӻ�ˮδ��������ζ�ʱ��ȥNaOH����Һ�����_______(�ƫ����ƫС������Ӱ�족)
�۵ζ�ʱ�ߵα�ҡ����ƿ���۾�Ӧ�۲�____________��
A���ζ�����Һ��ı仯 B����ƿ����Һ��ɫ�ı仯
�ܵζ��ﵽ�յ�ʱ����__________________________________________________��
��2���ζ�������±���ʾ��
| �ζ� ���� | ������Һ����� /mL | ����Һ�����/mL | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| 1 | 25��00 | 1��02 | 21��03 |
| 2 | 25��00 | 2��00 | 21��99 |
| 3 | 25��00 | 0��20 | 20��20 |
��NaOH����Һ��Ũ��Ϊ0��1010 mol��L��1�������Ʒ�е�����������Ϊ___________��
����6����ɫ��Һ���Ҵ������ӡ�Na2CO3��Һ��AgNO3��Һ��KOH��Һ�������ᣬ���ֵ��Լ���
| A�����Ƽ���Cu(OH)2����Һ | B��FeCl3��Һ |
| C��BaCl2��Һ | D������KMnO4��Һ |
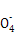 +8H+
+8H+ 5Fe3++Mn2++4H2O)��
5Fe3++Mn2++4H2O)��