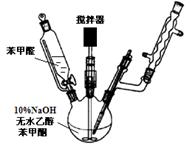
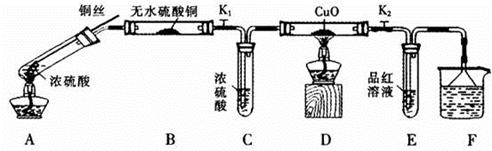
��Ŀ����
��ͭ��ʯ��Ҫ��������ͭ��Cu2S����������ʯ��SiO2����һ���Ի�ͭ��ʯΪԭ���Ʊ�����ͭ�Ĺ����������£�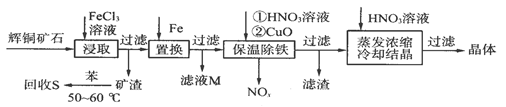
��д����ȡ������Cu2S�ܽ�����ӷ���ʽ��____________________��
�ƻ���S�������¶ȿ�����50��60��֮�䣬���˹�����͵�ԭ����_____________��
������NOx��������Ϻ�ͨ��ˮ�������������п�ѭ�����õ�һ�����ʣ��÷�Ӧ�Ļ�ѧ����ʽΪ___________________________������ҺM�м��루��ͨ�룩����__________������ĸ�����ʣ��õ���һ�ֿ�ѭ�����õ����ʡ�
a���� b������ c���������
�ȱ��³��������У�����CuO��Ŀ����________________________������Ũ��ʱ��Ҫ��HNO3��Һ������Һ��pH����������___________________________��
��1��Cu2S+4Fe3+=2Cu2++4Fe2++S
��2���¶ȹ��߱����ӷ����¶ȹ����ܽ�����С��
��3��4NOx+��5��2x��O2+2H2O= 4HNO3 b����4��������Һ��pH��ʹ��Ԫ�أ�Fe3+����ȫת��ΪFe(OH)3��������Cu2+��ˮ�⣨�������������ʣ�
����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����������ȷ����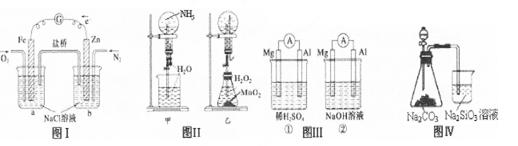
| A��ͼIװ����ͨ��N2��O2����������ͬ�� |
| B��ͼ���мס�����װ�ò�����Ȫ��ԭ��һ�� |
| C��ͼ���װ����þƬ��ԭ��صĸ�������װ����þƬ��ԭ��ص����� |
| D��ͼ����Һ©��ʢ������֤���ǽ�����N��C��Si��ʢ������֤���ǽ�����S��C��Si |
����ʵ����ȷ����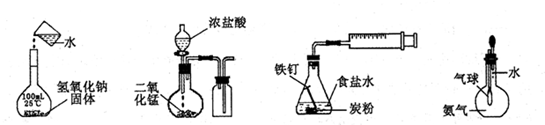
| A������һ��Ũ����Һ | B����ȡ���ռ�Cl2 | C���������ⸯʴ | D����֤����������ˮ |
(14��)
ij�о�С��Ϊ̽���������������������绯ѧ��ʴ���͵�Ӱ�����أ�����Ͼ��ȵ��������ۺ�̼��������ƿ�ײ�������ƿ��(��ͼ1)���ӽ�ͷ�ι��е��뼸�δ�����Һ��ͬʱ���������е�ѹǿ�仯��
��1�����������ʵ����Ʊ�(���в�Ҫ���ո�)��
| ��� | ʵ��Ŀ�� | ̼��/g | ����/g | ����/% |
| �� | Ϊ����ʵ�������� | 0.5 | 2.0 | 90.0 |
| �� | ����Ũ�ȵ�Ӱ�� | 0.5 | | 36.0 |
| �� | | 0.2 | 2.0 | 90.0 |
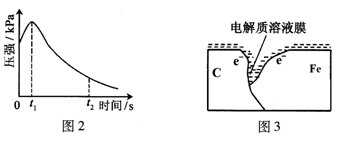
��3����С���ͼ2��0��t1ʱѹǿ����ԭ����������¼��裬������ɼ������
����һ���������ⸯʴ���������壻
������� ��
����
��4��Ϊ��֤����һ��ijͬѧ����˼����ռ����������Ƿ���H2�ķ��������������һ��ʵ�鷽����֤����һ��д��ʵ�鲽��ͽ��ۡ�
| ʵ�鲽��ͽ���(��Ҫ��д�����������)�� |
��14�֣�����34Se�����ڣ�52Te�����ǵ�VIA��Ԫ�أ����Ƿֲ��ڵؿ��е�ϡ��Ԫ�ء���ҵ�����ķ��ϣ���Ҫ�ɷ������ڡ�̼��ͭ�����Ͻ𣩻��վ��������������£�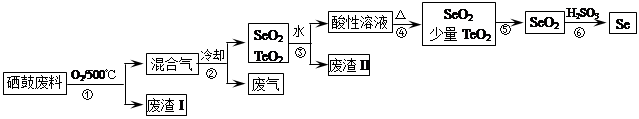
��֪��
| �������� | �۵� | �е� | ���� | �ܽ��� |
| SeO2 | 340�� | 684�� | 315�� | ������ˮ |
| TeO2 | 733�� | 1260�� | 450�� | ����ˮ |
��2���������ͨ�������ʹ���ķ��Ϸ��ڣ�Ŀ����______��
��3����������Ҫ�ɷ���______������II����Ҫ�ɷ���______��
��4�����������Ҫ��Ӧ�Ļ�ѧ����ʽ��______�������Ӧ�Ļ�ѧ����ʽ��______��
��5�����ݱ������ݣ�����������˵ķ��뷽����______��
��16�֣�ij��ѧ��ȤС���ڽ������ڿ�����ȼ�յ�ʵ�飬��д������������ȼ�յĻ�ѧ����ʽ�� ��ʵ���ͬѧ�Ƿ��ֳ��˵õ���ɫ�����⣬�����л��к�ɫ���ʡ���ȤС���ͬѧ�Բ����ĺ�ɫ�������ʽ���ʵ��̽����
�������ϣ�
��ҵ�ϲ����������������õ������ơ�ʵ�����н����Ƶı�ǩ��Ҫ�������£� 
������룺
����1���Ƹ���ú�ͣ�ú�͵IJ���ȫȼ�տ�������̼���ʣ�����2�� ���Ʒ�ӦҲ��������̼���ʣ�
����3�� �� ����4������
���ʵ�鷽������֤���룺
| ʵ��Ŀ�� | ʵ�鲽�� | ʵ������ | ���� |
| ��֤����1 | ��һС���ƴ�ú����ֱ��ȡ������ȼ�� ȡ��һС����ȥ��Ƥ�����ɾ�ú�͵��Ƽ���ȼ�� | | ��ɫ�����к� ��̼���� |
| ��֤����3 | | | ��ɫ������ ������Ԫ�� |
ͬѧ���˽��������ʯ������ȼ�գ���ʯ��������Ҳ�������ɺ����ĺ�ɫ���ʣ�������������ʯ������ȼ��̽�����ڿ�����ȼ�ղ����Ƿ������˵��ԭ�� ��
�����ʵ����֤����2����Ҫ��д����������̣��� ��
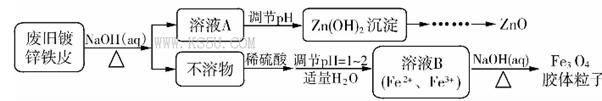
 Fe3O4�����壩+S4O62-+H2O
Fe3O4�����壩+S4O62-+H2O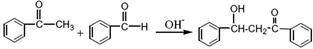
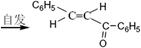 (���ͪ)
(���ͪ)