��Ŀ����
ij��ѧ��ȤС������ij����������ͭп����ȡ����ZnOʵ���������£�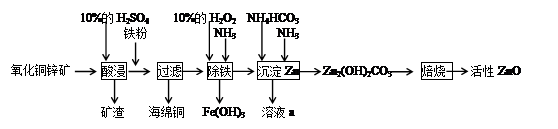
��ش��������⣺
��1���������ۺ�����Ӧ�����ӷ���ʽΪ_________________________________��
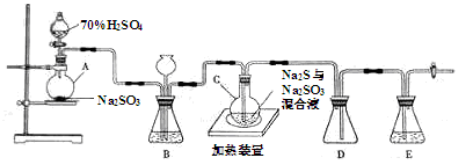
��2���ס�����ͬѧѡ���������������ò�ͬ�ķ�����ȡ������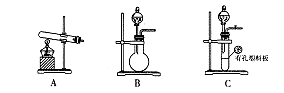
�ټ�ͬѧʹ�õ�ҩƷ����ʯ�����Ȼ�泥���Ӧѡ��װ��_______����дװ�ô��ţ������ɰ����Ļ�ѧ����ʽΪ____________________________________________________________________________��
����ͬѧѡ����װ��B����ʹ�õ�����ҩƷ������Ϊ_______________��
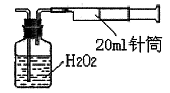
��3��H2O2��������____________________________________________________��
��4�����������еõ���Fe(OH)3����KClO��Һ�ڼ��Ի������������õ�һ�ָ�Ч�Ķ��ˮ��������K2FeO4�����÷�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ____________��
��5����֪��Һa�к���CO32����SO42��������������ӣ���ֻ����ȡ��һ����Ʒ�������������Ӵ��ڵ�ʵ���������Ϊ_______________________________________________________________________��
��1��Fe��2H����Fe2����H2����1�֣� Fe��Cu2����Fe2����Cu��1�֣�
��2����A��1�֣� 2NH4Cl��Ca(OH)2 CaCl2��2NH3��2H2O(2��)
CaCl2��2NH3��2H2O(2��)
��Ũ��ˮ��1�֣� ��ʯ�һ���ʯ�һ��������ƹ��壨2�֣�
��3������Һ�е�Fe2��������Fe3����2�֣� ��4��3:2��4�֣�
��5��ȡ������Һa���Թ��У��μ�BaCl2��Ba(NO3)2��Һ�����ˣ������ó����У��μӹ�������������ᣬ���������ܽ⣬˵����CO32����SO42����
���������������1�������������Һ������ͭ������п���������ᡣ���ǻ��õĽ�����������ǿ��ͭ�ģ�����п�ģ����Լ������ۺ�Ӧ�����ӷ���ʽΪFe��2H����Fe2����H2����Fe��Cu2����Fe2����Cu��
��2������ʯ�����Ȼ�臨��ǹ���ҩƷ������Ҫ��Һ©������Ӧѡ��װ����A����Ӧ�Ļ�ѧ����ʽΪ2NH4Cl��Ca(OH)2 CaCl2��2NH3��2H2O��
CaCl2��2NH3��2H2O��
�ڸ���Bװ�õ��ص��֪����װ�������ڹ�����Һ���Һ����Һ��֮�䲻��Ҫ�����Ʊ����壬�������ø�װ���Ʊ����������Һ©����ʢ�ŵ��Լ���Ũ��������ƿ��ʢ�ŵ��Ǽ�ʯ�һ���ʯ�һ��������ƹ��塣���ü�ʯ�һ���ʯ�һ��������ƹ�������ˮ������������Һ��OH��Ũ�ȣ��Ӷ��ٽ���ˮ��һˮ�ϰ��ֽ���ų�������
��3�����˺���Һ�к����������ӣ�Ҫת��Ϊ����������������Ҫ�����������������������ӣ����Լ���˫��ˮ�������ǽ���Һ�е�Fe2��������Fe3����
��4���������������������������ɸ�����أ�������Ԫ�صĻ��ϼ۴ӣ�3�����ߵ���6�ۣ�ʧȥ3�����ӡ�������������Ԫ�صĻ��ϼ۴ӣ�1�۽��͵���1�ۣ��õ�2�����ӡ���˸��ݵ��ӵ�ʧ�غ��֪����Ӧ���������뻹ԭ�������ʵ���֮����3:2��
��5������CO32����SO42��������������Ӷ��ܺ�Ba2���������̼�ᱵ�����ᱵ��ɫ��������̼�ᱵ���ܽ������У��ݴ˿��Լ����������ӡ�����Ϊֻ����ȡ��һ����Ʒ��������ȷ�IJ���������ȡ������Һa���Թ��У��μ�BaCl2��Ba(NO3)2��Һ�����ˣ������ó����У��μӹ�������������ᣬ���������ܽ⣬˵����CO32����SO42����
���㣺���鹤����������жϡ������Ʊ��ͼ��顢������ԭ��Ӧ���жϺ�Ӧ���Լ����ӵļ����

����ʵ��װ����̽��ͭ˿�����Ũ����ķ�Ӧ������������ȷ���� �� ��
| A�������ƶ�����ͭ˿�ɿ���SO2���� |
| B������Ʒ����Һ����ɫ |
| C������ʯ����Һ����ɫ |
| D��Ϊȷ��CuSO4���ɣ�����м�ˮ���۲���Һ��ɫ |
���л�ѧʵ�������ܹ��ﵽĿ�ĵ���
| A��Ϊ����KCl��AlCl3��MgCl2��Һ���ֱ���������Һ�еμ�NaOH��Һ������ |
| B������ȥ��������Һ�е�NaCl���ֲ��ı������ʣ��ɼ�������BaCl2��Һ����� |
| C������ˮ��pH�����ò�����պȡ��ˮ����pH��ֽ�ϣ������ɫ��ͱ���ɫ���Ƚ� |
| D��Ϊ��֤����¯���к����������ɽ���¯��ͨ�����ȵ�����ͭ��ĩ������ɫ��ĩ�Ƿ���ɫ |
������������(�ṹ��ʽ��ͼ)��һ�ּ�ǿ�����������Ȼ�����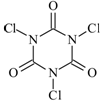
��1������������������ˮ������е�����������X�����������X�ķ���ʽΪ ��
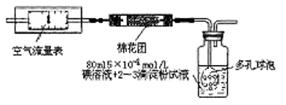
��2������Ч�ȡ�����ָ��KI����������ͬ����I2����Cl2��������ָ�������������֮�ȣ����ٷ�����ʾ��Ϊ�ⶨ������������ġ���Ч�ȡ��������ֳ�ȡij��������������Ʒ0.5680 g����ˮ������KI�����ᣬ���Ƴ�100 mL����Һ��ȷ��ȡ25.00 mL����Һ�ڵ���ƿ�У���0.1500 mol��L��1 Na2S2O3����Һ�ζ�����Һ����ɫʱ���������ָʾ���������ζ����յ�(������Ӧ�ķ���ʽΪ��2Na2S2O3��I2=Na2S4O6��2NaI)���ظ��ⶨ2�Σ����õ�����������±���
| �ζ���� | ����Һ���/mL | ��Һ�ζ���������/mL | ��Һ�ζ����յ����/mL |
| 1 | 25.00 | 0.06 | 24.04 |
| 2 | 25.00 | 0.02 | 24.02 |
| 3 | 25.00 | 0.12 | 24.14 |
�ٵζ��յ�۲쵽������Ϊ ��
������0.1500 mol��L��1 Na2S2O3��Һ100 mL������Na2S2O3��5H2O������Ϊ ��
�ۼ������Ʒ�ġ���Ч�ȡ�����(д���������)��
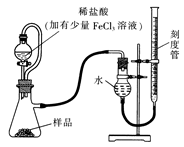
��ʽ̼����A������θҩ������ɿɱ�ʾΪAl2Mg6(OH)x(CO3)y��zH2O��ijУ��ѧ��ȤС�����ⶨ�仯ѧʽ��ʵ��������£�
ʵ��I����ȡһ��������A�����ȷֽ������ء�
ʵ���ȡһ��������A���������ᷴӦ����������CO2�����������
�ɹ�ѡ���������ҩƷ��ͼ��ʾ��������Һ��ѡ6mol/LHCl��6mol/LH2SO4�������Լ���ѡ����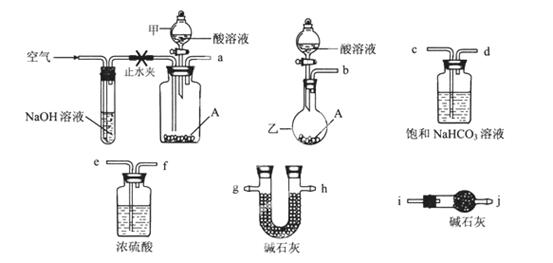
�ش��������⣺
(1)�����ҵ�����Ϊ________��
(2)��ѡ���Ҫ��װ�����ʵ��II,��ȷ������˳��Ϊ________ (�����������ýӿ���ĸ��ʾ����ѡ�õ�����Һ��________��
(3)�������������ʵ��I������ʵ��II��������A��ȫ��Ӧ��������Һ�еμ������İ�ˮ��������ֽ���ˣ�������ˮϴ�ӷ�Ӧ����2?3�Σ���ϴ��Һ���ˣ�ϴ�ӳ���2?3�Σ������ų�������ֽ�ŵ������м��ȷֽ������ء��жϳ�����ϴ�Ӹɾ��ķ�����_________________,ʵ����δ���ø÷�����ԭ���Dz�����ʵ����Ƶ�________ԭ������ĸ��ţ���
| A����ѧ�� | B����ȫ�� | C�������� | D����Լ�� |
(5)ͨ��ʵ��I��������ݣ�������ɵ�A��Ħ������Ϊ602.0g. mol-1��ʵ��II�гƵ���ƷA������Ϊ9.030g������������ȫ��Ӧ��CO2����װ������0.660g,��A �Ļ�ѧʽΪ________��
���и�ʵ��װ��ͼ�������У���ȷ���ǣ�������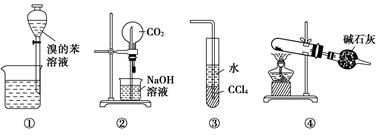
| A��װ�â�Ϊ�ų���ȡ��ˮ��ı��� |
| B��װ�â�Ϊ��Ȫʵ�� |
| C��װ�âۿ���������HCl���� |
| D����NH4ClΪԭ�ϣ�װ�âܿ������Ʊ�����NH3 |
 Na2S2O3��aq�� ��III��
Na2S2O3��aq�� ��III��