��Ŀ����
3��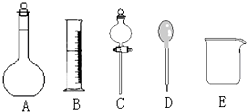 ��У������ȤС���ڴ�����ˮ��Ʒʱ��������������Ϊ36.5%��Ũ���ᣨ�ܶ�Ϊ1.19g/cm3�����Ƴ�240mL 0.1mol•L-1��������Һ��
��У������ȤС���ڴ�����ˮ��Ʒʱ��������������Ϊ36.5%��Ũ���ᣨ�ܶ�Ϊ1.19g/cm3�����Ƴ�240mL 0.1mol•L-1��������Һ����1������ͼ��ʾ�����У�����������Һ����Ҫ����C������ͼ��Ӧ��������ţ��� ��ͼ�����������⣬����������Һ����Ҫ�IJ��������Dz������������ð�ʹ�õ��Ⱥ�˳��ֱ��ǽ����������
��2�����ݼ��㣬��ҪŨ����2.1 mL���������̻����У���ʵ����ͲӦʹ�õ���A������ƿӦʹ��C�����ں�������д��Ӧ����ţ������֣���ͬ��
A��10mL B��100mL C��250mL D��500mL
��3������ȡŨ��������в����Ⱥ�˳���ǣ��ñ�ű�ʾ���ۢ٢ܢ�
�ٴ�ϡ�͵������¶�������һ�º��ز�����ע����ѡ����������ƿ�У�
��������ƿ��С�ļ�����ˮ��Һ��ӽ����ο̶���1��2cm�������ý�ͷ�ιܼ�����ˮ��ʹ��Һ����ʹ���ƿ���Ļ��ο̶������У�
����ʢ������ձ���ע����������ˮ�����ò�����������ʹ���Ͼ��ȣ�
��������ˮϴ���ձ��Ͳ�����2��3�Σ�����ϴ��Һȫ��ע������ƿ��
��4��������ƿ��ʹ�÷����У����в�����ȷ����AE��
A��ʹ������ƿǰ������Ƿ�©ˮ
B������ƿ��ˮϴ����������õ�ϡHCl��Һ��ϴ
C��������Һʱ����������ǹ��壬�ѳƺõ�������ֽ��С�ĵ�������ƿ�У�������ˮ���ӽ��̶���1��2cm�����ý�ͷ�ιܼ�����ˮ���̶���
D��������Һʱ����������Һ�壬����Ͳȡ������ֱ�ӵ�������ƿ�У�������ˮ���ӽ��̶���1��2cm�����ý�ͷ�ιܼ�����ˮ���̶���
E���Ǻ�ƿ������ʳָ��סƿ������һֻ����סƿ�ף�������ƿ������ת��Σ�ҡ��
��5����������ʱ������������ȷ��ֻ��������ijһ��������ж������Ƶ���ҺŨ�������Ҫ���ֵ����0.1mol/L����Σ���a��ƫ�ߣ�b��ƫ�ͣ�c����Ӱ�죬�����к���������Ӧ��ţ�
������ƿ������������ˮc
���ձ��Ͳ���û��ϴ��2-3��b
��ϡ��ŨHClʱ��û����ȴ������ת�Ƶ�����ƿ��a
�����Ƶ���Һװ��ྻ�ĵ�����������ˮ���Լ�ƿ��b
��������ʱ���ӣ�������Һ�����ʵ���Ũ��a��
���� ��1����������һ�����ʵ���Ũ�ȵ���Һʹ�õ�����������Ҫ��������ȱ�ٵ��������ٸ��ݲ������ڲ����е����ý��
��2������c=$\frac{1000�Ѧ�}{M}$�������ҪŨ�����Ũ�ȣ��ٸ�������250mL 0.1mol•L-1��������Һ��Ҫ���Ȼ�������ʵ����������Ҫ�����������250mL��Һ��Ҫ250mL����ƿ��
��3���������Ʋ����Ǽ��㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ���������Ʋ��裻
��4����������ƿ����ȷʹ�÷��������жϣ�
��5������c=$\frac{n}{V}$��������ʵ����ʵ���n����Һ�����V�ı仯��������������
��� �⣺��1����Һ©��������ȡ�ͷ�Һ������һ�����ʵ���Ũ����Һ���÷�Һ©������ѡC����ȱ�ٲ���������Ũ����ϡ��ʱ�ò��������裬ת��Һ��ʱ�ò�����������
�ʴ�Ϊ��C�������������裻������
��2����������Ϊ37%��Ũ���ᣨ�ܶ�Ϊ1.19g/cm3�������ʵ���Ũ��Ϊ��c=$\frac{1000�Ѧ�}{M}$=$\frac{1000��1.19��37%}{36.5}$=12.1��mol/L��������Ũ�������V=$\frac{0.1mol/L��0.25L}{12.1mol/L}$=0.0021L=2.1mL����ѡ��10mL��Ͳ������250mL��Һ��Ӧѡ��250mL������ƿ��
�ʴ�Ϊ��2.1��A��C��
��3���������Ʋ����Ǽ��㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��֪��ȷ�IJ��������Ǣۢ٢ܢڣ��ʴ�Ϊ���ۢ٢ܢڣ�
��4��A������ƿʹ��ʱ��Ӧ�ȼ���Ƿ�©ˮ��Ȼ��������ˮϴ�Ӹɾ����ɣ���A��ȷ��
B������ƿϴ����������������Һ��ϴ������Ӱ�����Ƶ���Һ��Ũ�ȣ���B����
C������ƿֻ������������Һ������������ƿ���ܽ⣬Ӧ�����ձ����ܽ⣬��C����
D������ƿֻ������������Һ������������ƿ��ϡ�ͣ�Ӧ�����ձ���ϡ�ͣ���D����
E��ҡ��ʱ���Ǻ�ƿ������ʳָ��סƿ������һֻ����סƿ�ף�������ƿ������ת����E��ȷ��
��ѡAE��
��5��������ƿ������������ˮ����������Һ��Ũ����Ӱ�죬��ѡc��
���ձ��Ͳ���û��ϴ��2-3�Σ���ᵼ�����ʵ���ʧ��Ũ��ƫ�ͣ���ѡb��
��ϡ��ŨHClʱ��û����ȴ������ת�Ƶ�����ƿ�У�����ȴ����Һ���ƫС��Ũ��ƫ�ߣ���ѡa��
�����Ƶ���Һװ��ྻ�ĵ�����������ˮ���Լ�ƿ�У������Һ���ϡ�ͣ���Ũ��ƫ�ͣ���ѡb��
��������ʱ���ӣ�����Һ���ƫС��������Һ�����ʵ���Ũ��ƫ�ߣ���ѡa��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���Ŀ�ѶȲ���Ҫ��ѧ��������������һ�����ʵ���Ũ�ȵ���Һ�ķ�������ɷ���������ע��������ɸ���c=$\frac{n}{V}$���з����������ʵ����ʵ���n�������ƽ��ƫ�ߣ�����Һ�����V���������Ƶ���ҺŨ��ƫ�ͣ�

| A�� | 5.6������Ӧת�Ƶĵ�����һ��Ϊ3NA | |
| B�� | 1mol Cl2��Ӧת�Ƶĵ�����һ����2NA | |
| C�� | ��״���£�22.4L SO3���е���ԭ����ĿΪ3NA | |
| D�� | 1mol̼������CH5+�����ĵ�������Ϊ10NA |
| A�� | ��ˮ�� | B�� | ǿ���� | C�� | ��ˮ�� | D�� | ǿ������ |
| A�� | $\frac{a+b}{4}$ | B�� | $\frac{3a+2b}{4}$ | C�� | 4��a+b�� | D�� | 4��3a+b�� |
| A�� | ƽ�ⳣ����С | B�� | ����A��ת���������� | ||
| C�� | ƽ��������Ӧ�����ƶ��� | D�� | a��b |
| A�� | ����ֱͨ���罺���������ƶ����ǵ������Һֱͨ�������ʲ��ƶ� | |
| B�� | ��Һ������������ͨ����ֽ�������з�ɢ�����Ӳ���ͨ����ֽ | |
| C�� | ��Һ��ͨ��һ������û��������������ͨ��һ�����߳������Թ�� | |
| D�� | ��Һ�ȶ������ú����ɳ��������岻�ȶ������û����ɳ��� |
| A�� | �ڲⶨ��Һ��pHʱ����������ˮʪ���ò�������ȡ��Һ������ֽ�в����������ɫ���Ƚ� | |
| B�� | ij��Һ��BaCl2��Һ���ɰ�ɫ������˵��ԭ��Һ����SO42- | |
| C�� | �ᴿ��������Ba��NO3��2���ʵ�KNO3��Һ������ʹ�õķ���Ϊ���������K2CO3��Һ�����˳�ȥ������������Һ�в�������HNO3 | |
| D�� | ʵ��������ò�����ƺͰ���Ӧֱ���ӵ���Һ���� |
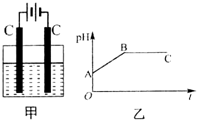 ��װ���������������ʵ���֮��Ϊ1��2��CuSO4��NaCl�Ļ����Һ������������Һ��pHֵ��ʱ��t�仯��ʾ��ͼ����ʾ�������ǵ����������ˮ�ķ�Ӧ�����Է���������������ȷ���ǣ�������
��װ���������������ʵ���֮��Ϊ1��2��CuSO4��NaCl�Ļ����Һ������������Һ��pHֵ��ʱ��t�仯��ʾ��ͼ����ʾ�������ǵ����������ˮ�ķ�Ӧ�����Է���������������ȷ���ǣ�������| A�� | �Ǹû����Һ�е�SO42-������A����Һ��pHֵС��B�� | |
| B�� | BC�����������������������֮��Ϊ2��1 | |
| C�� | AB�߶���BC�߶��������Ϸ����ķ�Ӧ����ͬ�ļ���Cu2++2e-��Cu | |
| D�� | ���������Ĺ����л������������ɫ��Cu��OH��2���� |
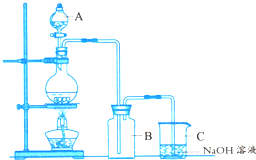 ʵ���ҿ����ö������̺�Ũ�����ڼ����������Ʊ���������ͼΪ
ʵ���ҿ����ö������̺�Ũ�����ڼ����������Ʊ���������ͼΪ