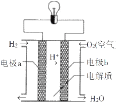
ЬтФПФкШн
ЁОЬтФПЁПЮэіВгЩЖржжЮлШОЮяаЮГЩ,АќКЌПХСЃЮяЃЈPM2.5ЃЉЁЂЕЊбѕЛЏЮяЃЈNOxЃЉЁЂCOЁЂSO2ЕШЁЃЛЏбЇдкНтОіЮэіВЮлШОжагазХживЊЕФзїгУЁЃ
ЃЈ1ЃЉвбжЊ:Ђй![]() ІЄH1=-566.0 kJЁЄmol-1Ђк
ІЄH1=-566.0 kJЁЄmol-1Ђк![]() ЁЁІЄH2=-116.5 kJЁЄmol-1Ђл
ЁЁІЄH2=-116.5 kJЁЄmol-1Ђл![]() ЁЁ ІЄH3=+180.5 kJЁЄmol-1ЃЌЗЯЦјжаNO2 гыCO зЊЛЏГЩЮоЮлШОЦјЬхЕФШШЛЏбЇЗНГЬЪНЮЊ____ЁЃ
ЁЁ ІЄH3=+180.5 kJЁЄmol-1ЃЌЗЯЦјжаNO2 гыCO зЊЛЏГЩЮоЮлШОЦјЬхЕФШШЛЏбЇЗНГЬЪНЮЊ____ЁЃ
ЃЈ2ЃЉбаОПЗЂЯжРћгУNH3ПЩГ§ШЅЯѕЫсЙЄвЕЮВЦјжаЕФNOЁЃNH3гыNOЕФЮяжЪЕФСПжЎБШЗжБ№ЮЊ1ЁУ2ЁЂ1ЁУ1.5ЁЂ3ЁУ1ЪБ,NOЭбГ§ТЪЫцЮТЖШБфЛЏЕФЧњЯпШчЭМЫљЪОЁЃ

ЂйЧњЯпaжа,NOЕФЦ№ЪМХЈЖШЮЊ6ЁС10-4mgЁЄm-3,ДгXЕуЕНYЕуОЙ§10 s,дђИУЪБМфЖЮФкNOЕФЭбГ§ЫйТЪЮЊ___________mgЁЄ m-3ЁЄs-1ЁЃ
ЂкЧњЯпcЖдгІЕФNH3гыNOЕФЮяжЪЕФСПжЎБШЪЧ___,ЦфРэгЩЪЧ___ЁЃ
ЃЈ3ЃЉЬПКкЪЧЮэіВжаЕФживЊПХСЃЮя,баОПЗЂЯжЫќПЩвдЛюЛЏбѕЗжзг,ЩњГЩЛюЛЏбѕ,ЛюЛЏбѕПЩвдПьЫйбѕЛЏSO2ЁЃЛюЛЏЙ§ГЬЕФФмСПБфЛЏФЃФтМЦЫуНсЙћШчЭМЫљЪОЁЃ
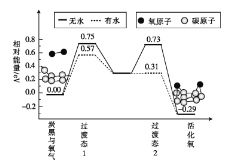
ЮоЫЎЧщПіЯТ,вЛИібѕЗжзгЕФЛюЛЏФмЮЊ__ЁЁ,ШнвзЛюЛЏбѕЗжзгЕФЬѕМўЪЧ___ЃЈЬюЁАгаЫЎЁБЛђЁАЮоЫЎЁБЃЉЁЃ
ЃЈ4ЃЉРћгУЗДгІ![]() ЙЙГЩЕФЕчГиМШФмгааЇЯћГ§ЕЊбѕЛЏЮяЕФХХЗХ,МѕЧсЮэіВЮлШО,гжФмГфЗжРћгУЛЏбЇФм,зАжУШчЭМЫљЪОЁЃ
ЙЙГЩЕФЕчГиМШФмгааЇЯћГ§ЕЊбѕЛЏЮяЕФХХЗХ,МѕЧсЮэіВЮлШО,гжФмГфЗжРћгУЛЏбЇФм,зАжУШчЭМЫљЪОЁЃ
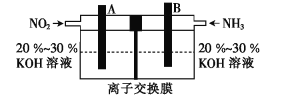
ЂйBМЋЕФЕчМЋЗДгІЪНЮЊ___ЁЃ
ЂкШєЗДгІзЊвЦ1.2 molЕчзг,AМЋЩњГЩN2ЕФЬхЛ§ЮЊ___LЃЈБъзМзДПіЃЉЁЃ
ЁОД№АИЁП2NO2ЃЈgЃЉ+4COЃЈgЃЉ=N2ЃЈgЃЉ+4CO2ЃЈgЃЉ ЁїH=ЁЊ1196.0kJЁЄmol-1 1.2ЁС10ЁЊ5 1ЃК2 NH3гыNOЕФЮяжЪЕФСПЕФБШжЕдНДѓЃЌNOЭбГ§ТЪдНДѓ 0.75eV гаЫЎ 2NH3-6e-+6OH-=N2+6H2O 3.36
ЁОНтЮіЁП
ЃЈ1ЃЉгЩИЧЫЙЖЈТЩПЩжЊЃЌЂйЁС2ЁЊЂкЁЊЂлПЩЕУЗЯЦјжаNO2гыCO зЊЛЏГЩЮоЮлШОЦјЬхЕФШШЛЏбЇЗНГЬЪНЮЊ2NO2ЃЈgЃЉ+4COЃЈgЃЉ=N2ЃЈgЃЉ+4CO2ЃЈgЃЉЃЌдђЁїH=ЁїH1ЁС2ЁЊЁїH2ЁЊЁїH3=ЃЈ-566.0 kJЁЄmol-1ЃЉЁЊЃЈ-116.5 kJЁЄmol-1ЃЉЁЊЃЈ+180.5 kJЁЄmol-1ЃЉ=ЁЊ1196.0kJЁЄmol-1ЃЌЙЪД№АИЮЊЃК2NO2ЃЈgЃЉ+4COЃЈgЃЉ=N2ЃЈgЃЉ+4CO2ЃЈgЃЉ ЁїH=ЁЊ1196.0kJЁЄmol-1ЃЛ
ЃЈ2ЃЉЂйЧњЯпaжаЃЌNOЕФЦ№ЪМХЈЖШЮЊ6ЁС10-4mg/m3ЃЌAЕуЕФЭбГ§ТЪЮЊ0.55ЃЌBЕуЕФЭбГ§ТЪЮЊ0.75ЃЌДгAЕуЕНBЕуОЙ§10sЃЌИУЪБМфЖЮФкNOЕФЭбГ§ЫйТЪ=![]() =1.2ЁС10ЁЊ5 mgЁЄ m-3ЁЄs-1ЃЌЙЪД№АИЮЊЃК1.2ЁС10ЁЊ5ЃЛ
=1.2ЁС10ЁЊ5 mgЁЄ m-3ЁЄs-1ЃЌЙЪД№АИЮЊЃК1.2ЁС10ЁЊ5ЃЛ
ЂкNH3гыNOЕФЮяжЪЕФСПЕФБШжЕдНДѓЃЌNH3ЕФЮяжЪЕФСПдНДѓЃЌNOЭбГ§ТЪдНДѓЃЌЮяжЪЕФСПжЎБШЗжБ№ЮЊ1ЃК2ЁЂ1ЃК1.5ЁЂ3ЃК1ЫљЖдгІЕФЧњЯпЗжБ№ЮЊcЁЂbЁЂaЃЌЙЪЧњЯпcЖдгІNH3гыNOЕФЮяжЪЕФСПжЎБШЪЧ1ЃК2ЃЌЙЪД№АИЮЊЃК1ЃК2ЃЛNH3гыNOЕФЮяжЪЕФСПЕФБШжЕдНДѓЃЌNOЭбГ§ТЪдНДѓЃЛ
ЃЈ3ЃЉИљОнФмСПЭМЗжЮіЃЌећИіЗДгІЕФЛюЛЏФмЮЊЛюЛЏФмНЯДѓепЃЌдђУЛгаЫЎМгШыЕФЗДгІЛюЛЏФмЮЊE=0.75eVЃЛгаЫЎМгШыЕФЗДгІЕФЛюЛЏФмЮЊE=0.57eVЃЌЫљвдЫЎПЩЪЙбѕЗжзгЛюЛЏЗДгІЕФЛюЛЏФмНЕЕЭ0.75eV-0.57eV=0.18eVЃЌЙЪД№АИЮЊЃК0.75eVЃЛгаЫЎЃЛ
ЃЈ4ЃЉЂйИљОнзАжУЭМПЩжЊЃЌЕчМЋBЭЈШыАБЦјЃЌдкKOHШмвКжаАБЦјБЛбѕЛЏЮЊЕЊЦјЃЌЕчМЋЗДгІЪНЮЊ2NH3-6e-+6OH-=N2+6H2OЃЌЙЪД№АИЮЊЃК2NH3-6e-+6OH-=N2+6H2OЃЛ
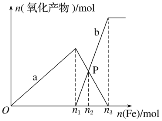
ЂкгЩЛЏбЇЗНГЬЪНПЩжЊЃЌAМЋЯћКФ5molЖўбѕЛЏЕЊЃЌЩњГЩ3molЕЊЦјЃЌЗДгІзЊвЦ24molЕчзгЃЌдђЗДгІзЊвЦ1.2 molЕчзгЃЌAМЋЩњГЩ0.15molЕЊЦјЃЌБъзМзДПібЁЬхЛ§ЮЊ0.15molЁС22.4L/mol=3.36LЃЌЙЪД№АИЮЊЃК3.36ЁЃ

 ПьРжаЁВЉЪПЙЎЙЬгыЬсИпЯЕСаД№АИ
ПьРжаЁВЉЪПЙЎЙЬгыЬсИпЯЕСаД№АИЁОЬтФПЁПФГЛЏбЇаЫШЄаЁзщЮЊСЫЬНОПТСЕчМЋдкЕчГижаЕФзїгУЃЌЩшМЦВЂНјааСЫвдЪЕбщНсЙћМЧТМШчЯТ:
БрКХ | ЕчМЋВФСЯ | ЕчНтжЪШмвК | ЕчСїБэжИеыЦЋзЊЗНЯђ |
1 | AlЁЂCu | ЯЁбЮЫс | ЦЋЯђCu |
2 | AlЁЂCЃЈЪЏФЋЃЉ | ЯЁбЮЫс | ЦЋЯђЪЏФЋ |
ЛиД№ЯТСаЃЈ1ЃЉЁЂЃЈ2ЃЉаЁЬт:
ЂХЪЕбщ1жаAlзїЕФЕчМЋЮЊ____(ЬюЁАе§МЋЁБЛђЁАИКМЋЁБ)ЃЌCuзїЕФЕчМЋЮЊ____(ЬюЁАе§МЋ"ЛђЁАИКМЋЁБ)ЁЃ
ЂЦЪЕбщ2ЙлВьЕНC(ЪЏФЋ)АєЩЯВњЩњЕФЯжЯѓЪЧ____,ИКМЋЕФЕчМЋЗДгІЪНЮЊ____.ШчЭМЪЧЕчНтжЪЮЊЯЁСђЫсШмвКЕФЧтбѕШМСЯЕчГиЁЃИУЕчГиЕФЕчзгДгЕчМЋ____ОЙ§ЕМЯпСїЯђЕчМЋ____(ЬюaЛђb)