��Ŀ����
�����ʽṹ�����ʡ�
�Ե�����(GaN)Ϊ�����ĵ������뵼�����Ŀǰ�ѳ�Ϊȫ��뵼���о���ǰ�غ��ȵ㡣�ش��������⣺
��1����ΪԪ�����ڱ���31��Ԫ�أ���ԭ�Ӽ۲�����Ų�ͼΪ___________��
��2�������������е�һ����������Ԫ����________����Ԫ�ط��ţ���ͬ���������������е縺������Ԫ����____________________��
��3����ͳ�ĵ������Ʊ������Dz���GaCl3��NH3��һ�������·�Ӧ���÷�Ӧ�Ļ�ѧ����ʽΪ______________��
��4������������ʯ�������Ƶľ���ṹ���������е�ԭ������ԭ��֮����____���ϣ�����������_______���塣
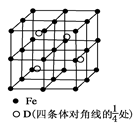
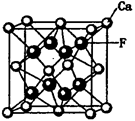
��5����ͼ�ǵ����صľ���ģ�ͣ�
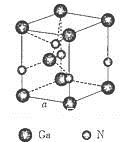
�ٵ���������ԭ�ӵ��ӻ���ʽΪ__________����ԭ�ӵ���λ��Ϊ___________��
�ڵ�����Ϊ���������������ص��ܶ�Ϊd g/cm3���г����㵪���ؾ����߳�a�ı���ʽ��a=_______cm��
�Ե�����(GaN)Ϊ�����ĵ������뵼�����Ŀǰ�ѳ�Ϊȫ��뵼���о���ǰ�غ��ȵ㡣�ش��������⣺
��1����ΪԪ�����ڱ���31��Ԫ�أ���ԭ�Ӽ۲�����Ų�ͼΪ___________��
��2�������������е�һ����������Ԫ����________����Ԫ�ط��ţ���ͬ���������������е縺������Ԫ����____________________��
��3����ͳ�ĵ������Ʊ������Dz���GaCl3��NH3��һ�������·�Ӧ���÷�Ӧ�Ļ�ѧ����ʽΪ______________��
��4������������ʯ�������Ƶľ���ṹ���������е�ԭ������ԭ��֮����____���ϣ�����������_______���塣
��5����ͼ�ǵ����صľ���ģ�ͣ�

�ٵ���������ԭ�ӵ��ӻ���ʽΪ__________����ԭ�ӵ���λ��Ϊ___________��
�ڵ�����Ϊ���������������ص��ܶ�Ϊd g/cm3���г����㵪���ؾ����߳�a�ı���ʽ��a=_______cm��
��1��
��2��N ��B
��3��GaCl3+NH3=GaN+3HCl
��4�����ۼ� ԭ��
��5����sp2 4 ��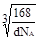

��2��N ��B
��3��GaCl3+NH3=GaN+3HCl
��4�����ۼ� ԭ��
��5����sp2 4 ��
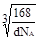
��1����ԭ������Ϊ31���������������Ų�ʽΪ��1s22s22p63s23p63d104s24p1����������Ϊ�۵��ӣ��۵����Ų�ʽΪ��4s24p1����4s�����һ�������෴�ĵ��ӣ�4p���ֻ��һ�����ӣ��ʴ�Ϊ�� ��
��
��2����һ������ͬ������ϵ��£�Խ��ԽС��NԪ��Ϊ�������������Ԫ�أ���һ��������縺�Դ��ϵ��£�Խ��ԽС�����������������Ԫ��ΪB��
��3����Ӧ��ΪNH3��GaCl3��������ΪGaN�������жϳ���һ�ֲ���ΪHCl������ԭ���غ�д����ѧ����ʽ���ʴ�Ϊ��GaCl3+NH3=GaN+3HCl��
��4�����ڵ���������ʯ�������Ƶľ���ṹ�����Ե�����Ϊԭ�Ӿ��壬ԭ��֮���Թ��ۼ������һ��
��5���ٸ��ݾ������Կ���Ga�������ڵ�����N�γɹ��ۼ�����Ga�γ��������ۼ��������ӻ�����Ϊsp2�ӻ����۲쾧���ṹ����Nԭ����Χ���������Ga��ĿΪ4������λ��Ϊ4��
��GaN�����У�Gaλ�ڶ�������ģ����Ժ���Ga��Ϊ��8��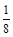 +1=2��Nԭ��λ��������ģ�����N��Ϊ��4��
+1=2��Nԭ��λ��������ģ�����N��Ϊ��4��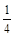 +1=2��GaN�����к�������GaN�������߳�Ϊ
+1=2��GaN�����к�������GaN�������߳�Ϊ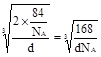
 ��
����2����һ������ͬ������ϵ��£�Խ��ԽС��NԪ��Ϊ�������������Ԫ�أ���һ��������縺�Դ��ϵ��£�Խ��ԽС�����������������Ԫ��ΪB��
��3����Ӧ��ΪNH3��GaCl3��������ΪGaN�������жϳ���һ�ֲ���ΪHCl������ԭ���غ�д����ѧ����ʽ���ʴ�Ϊ��GaCl3+NH3=GaN+3HCl��
��4�����ڵ���������ʯ�������Ƶľ���ṹ�����Ե�����Ϊԭ�Ӿ��壬ԭ��֮���Թ��ۼ������һ��
��5���ٸ��ݾ������Կ���Ga�������ڵ�����N�γɹ��ۼ�����Ga�γ��������ۼ��������ӻ�����Ϊsp2�ӻ����۲쾧���ṹ����Nԭ����Χ���������Ga��ĿΪ4������λ��Ϊ4��
��GaN�����У�Gaλ�ڶ�������ģ����Ժ���Ga��Ϊ��8��
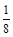 +1=2��Nԭ��λ��������ģ�����N��Ϊ��4��
+1=2��Nԭ��λ��������ģ�����N��Ϊ��4��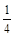 +1=2��GaN�����к�������GaN�������߳�Ϊ
+1=2��GaN�����к�������GaN�������߳�Ϊ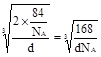

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ


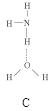
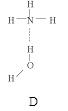
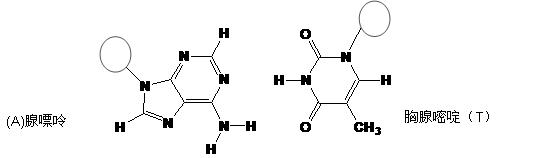
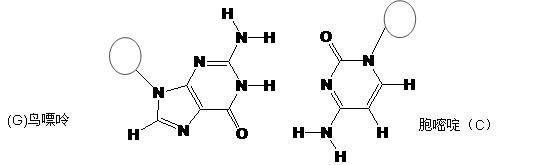
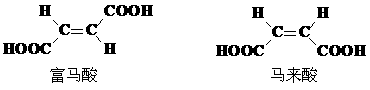

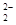 ���ڣ�ʹ������һ��������������CaC2������1��C
���ڣ�ʹ������һ��������������CaC2������1��C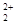 ��Ϊ�ȵ����壬O
��Ϊ�ȵ����壬O