��Ŀ����
����Ŀ��A���ɵ��Ȳ����Ƴɵ��ܱ�������B��һ�ͻ�ѧ��ʴ�����ڴ��ȵ����ر�K2����������������NO2ͨ��K1��K3�ֱ����A��B�У���Ӧ��ʼʱ��A��B�������ͬ��(��֪��2NO2(g) ![]() N2O4(g)����H��0)
N2O4(g)����H��0)
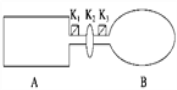
��1��һ��ʱ���Ӧ�ﵽƽ�⣬��ʱA��B������N2O4��������vA______vB(������������������������)��������K2������B��______(���������������С������������)��
��2���رջ���K2������A��B���ٳ������ʼ����ȵ�NO2����ﵽƽ��ʱ��NO2��ת������A��________(����������������С������������)�����ֱ�ͨ���������������ﵽƽ��ʱ��A��NO2��ת���ʽ�________��B��NO2��ת���ʽ�______(���������������С������������)��
��3�������£���A��B������������䣬��A����һ�����Ȳ㣬B�������Խ����ȴ��ݣ���ﵽƽ��ʱ��______�е���ɫ���
��4����������A�г���4.6 g��NO2���ﵽƽ��������ڻ�������ƽ����Է�������Ϊ57.5����ƽ��ʱN2O4�����ʵ���Ϊ___________________��
���𰸡� �� ��С ���� ���� ��С A 0.02mol
�������������������1������װ�ÿ�֪��A�DZ��ֺ��º��ݵģ�B�DZ��ֱ��ֺ��º�ѹ�ġ����ڸ÷�Ӧ�������С�ķ��ȵĿ��淴Ӧ������A�е�ѹǿ�ڷ�Ӧ�����м�С������A�еķ�Ӧ����С��B�еķ�Ӧ���ʡ�������K2�����൱������װ���Ǻ��º�ѹ�ģ���������B����С����2���ڼ��������NO2���壬��A���൱������ѹǿ��ƽ��������Ӧ�����ƶ���ת����������ͨ�������Ne������A�����ʵ�Ũ�Ȳ��䣬ƽ�ⲻ�ƶ���ת���ʲ��䣻��B��ѹǿ����ģ����������ݻ��������ʵ�Ũ�ȼ�С��ƽ�����淴Ӧ������У�ת���ʼ�С����3���÷�ӦΪ���ȷ�Ӧ����A�����������Ȳ㣬�൱�ڸ�A���ȣ�ƽ�������ƶ���NO2��Ũ������A�е���ɫ���
��4��4��6g��NO2�����ʵ���Ϊ0��1mol,n(��)=4��6/57��6=0��08mol,��������ʽ���⣺
2NO2![]() N2O4
N2O4
��ʼ 0��1 0
�仯 2x x
ƽ��0��1-2x x 0��1-2x+x=0��08 x=0��02mol








