��Ŀ����
A��B��C��D��E��F���ֶ�����Ԫ�ص�ԭ����������������֪����Aԭ�Ӱ뾶��̣���Bԭ�������������Ǵ�����������2������Cԭ�������������ȴ�����������4������Dԭ�ӵĴ�����������������������8������E���ʼ��ܺ����ᷴӦ���ܺ��ռӦ����F��Cͬ���塣�ش��������⣺
��1��������Ԫ���У��γ���������������______����Ԫ�����ƣ���
��2��д��CԪ�����е��⻯��ĵ���ʽ______��
��3���õ���ʽ��ʾD2F���γɹ���______��
��4��A2C�ķе��A2F�ķе�ߵ���Ҫԭ��______��
��5��д��E���ռ���Һ��Ӧ�����ӷ���ʽ______��
��1��������Ԫ���У��γ���������������______����Ԫ�����ƣ���
��2��д��CԪ�����е��⻯��ĵ���ʽ______��
��3���õ���ʽ��ʾD2F���γɹ���______��
��4��A2C�ķе��A2F�ķе�ߵ���Ҫԭ��______��
��5��д��E���ռ���Һ��Ӧ�����ӷ���ʽ______��
��12�֣���1��̼��2�֣�

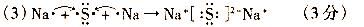
��4��ˮ���Ӽ������� ��2�֣�
��5��2Al+2OH��+2H2O=2AlO2��+3H2 ��2�֣�������������ͬ�����֣�
��2�֣�������������ͬ�����֣�

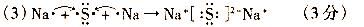
��4��ˮ���Ӽ������� ��2�֣�
��5��2Al+2OH��+2H2O=2AlO2��+3H2
 ��2�֣�������������ͬ�����֣�
��2�֣�������������ͬ�����֣�����Ԫ�صĽṹ��Ӧ�����ʿ�֪�� A��B��C��D��E��F���ֶ�����Ԫ��H��C��O��Na��Al��S��
��1�����������������л�����л��ﶼ�Ǻ���̼Ԫ�ء�
��2����Ԫ�����γ������⻯��ˮ��˫��ˮ������ʽ�ֱ���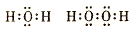 ��
��
��3�������Ǻ������Ӽ������ӻ�������γɹ��̿ɱ�ʾΪ
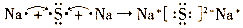 ��
��
��4������ˮ�����к������������ˮ�ķе��������ġ�
��5����������������Һ��Ӧ����������ƫ�����ƣ�����ʽΪ2Al+2OH��+2H2O=2AlO2��+3H2����
��1�����������������л�����л��ﶼ�Ǻ���̼Ԫ�ء�
��2����Ԫ�����γ������⻯��ˮ��˫��ˮ������ʽ�ֱ���
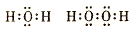 ��
����3�������Ǻ������Ӽ������ӻ�������γɹ��̿ɱ�ʾΪ
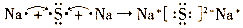 ��
����4������ˮ�����к������������ˮ�ķе��������ġ�
��5����������������Һ��Ӧ����������ƫ�����ƣ�����ʽΪ2Al+2OH��+2H2O=2AlO2��+3H2����

��ϰ��ϵ�д�
�����Ŀ
 ��
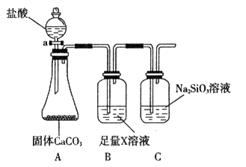
��
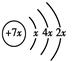 ��B��ͬ���ڳ�ϡ��������뾶����Ԫ�أ�C��������������ɵ����ӣ�E��F����Χ�����Ų�ʽ�ֱ�Ϊ3d54s1��3d64s2���ش��������⣺
��B��ͬ���ڳ�ϡ��������뾶����Ԫ�أ�C��������������ɵ����ӣ�E��F����Χ�����Ų�ʽ�ֱ�Ϊ3d54s1��3d64s2���ش��������⣺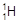 ��
��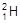 ��
�� ��H+��H2���Ա�ʾ �� ��
��H+��H2���Ա�ʾ �� ��