��Ŀ����
5�������Ԫ�����ڱ���һ���֣���Ա��еĢ�-����Ԫ�أ���д���пհף�| ���� | IA | ��A | ��A | IVA | VA | VIA | V��A | 0�� |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� |
��2��������������ˮ�����У�������ǿ�Ļ�����ķ���ʽ��HClO4��������ǿ�Ļ�����ĵ���ʽ�ǣ�
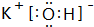 ��
����3������������������������Ԫ����Al����Ԫ�ط��ţ���д���������������������Ʒ�Ӧ�����ӷ���ʽAl2O3+2OH-=2AlO2-+H2O��
��4���õ���ʽ��ʾԪ�آ���Ļ�������γɹ���
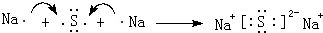 ��
��
���� ��Ԫ�������ڱ��е�λ�ÿ�֪����ΪC����ΪN����ΪO����ΪNa����ΪAl����ΪS����ΪCl����ΪAr����ΪK��
��1��ϡ�����������Ϊ�ȶ��ṹ����ѧ��������ã�
��2���ǽ�����Խǿ����ۺ����������Խǿ������������ˮ�����У�������ǿ�Ļ�����ΪKOH���������ӻ�����ɼ����������������ӹ��ɣ�
��3������������������������������Ʒ�Ӧ����ƫ��������ˮ��
��4��Ԫ�آ���Ļ�����ΪNa2S�����������������ӹ��ɣ���Naԭ�ӡ�Sԭ�ӵ���ʽ��ʾ���γɹ��̣�
��� �⣺��Ԫ�������ڱ��е�λ�ÿ�֪����ΪC����ΪN����ΪO����ΪNa����ΪAl����ΪS����ΪCl����ΪAr����ΪK��
��1��ϡ������Arԭ�������Ϊ�ȶ��ṹ����ѧ��������ã��ʴ�Ϊ��Ar��
��2���ǽ�����Խǿ����ۺ����������Խǿ����HClO4��������ǿ������������ˮ�����У�������ǿ�Ļ�����ΪKOH���������ӻ�����ɼ����������������ӹ��ɣ�����ʽΪ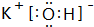 ��
��
�ʴ�Ϊ��HClO4��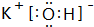 ��
��
��3��AlԪ�ص�������--������������������������������Ʒ�Ӧ����ƫ��������ˮ����Ӧ���ӷ���ʽΪ��Al2O3+2OH-=2AlO2-+H2O��
�ʴ�Ϊ��Al��Al2O3+2OH-=2AlO2-+H2O��
��4��Ԫ�آ���Ļ�����ΪNa2S�����������������ӹ��ɣ���Naԭ�ӡ�Sԭ�ӵ���ʽ��ʾ���γɹ���Ϊ��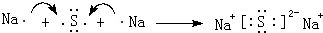 ��
��
�ʴ�Ϊ��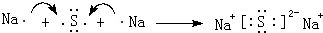 ��
��
���� ���⿼��ѧ��Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ�ã����س��û�ѧ���Ԫ�������ɵĿ��飬�ѶȲ���ע�����֪ʶ���������գ�

| A�� | ���������ʻ��ã�����������Ϻ�����������ը | |
| B�� | ����HCl�������Ƿ����Cl2�����ǽ�����ͨ����������Һ | |
| C�� | ŨHCl����MnO2������ȡCl2��ʵ����ֻ��ԭ�������� | |
| D�� | ʵ������ȡ����ʱ��Ϊ�˷�ֹ������Ⱦ���������������������������Һ���� |
| A�� | ���л��ﺬ�����ֹ����� | |
| B�� | ���л������ڷ����� | |
| C�� | ���л���ɷ���ȡ�����ӳɡ��Ӿۡ���������ԭ����ȥ��Ӧ | |
| D�� | 1 mol���л������ʵ������£�������5 molH2��Ӧ |
| A�� | �Ҵ�������������Ʒ�Ӧ�ų����� | |
| B�� | ��ϩ�ͱ�����ʹ��ˮ��ɫ����ԭ����ͬ | |
| C�� | �����ʵ�������������춡�飬���зǼ��Լ�����Ŀ��ͬ | |
| D�� | �����ڹ�����������Cl2����ȡ����Ӧ�����õ�5�ֲ��� |
| A�� | ��ҵұ����Fe2O3+3CO$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2 | |
| B�� | ����ͭCu2S+O2$\frac{\underline{\;����\;}}{\;}$2Cu+SO2 | |
| C�� | ��ҵұ��þMgCl2$\frac{\underline{\;���\;}}{\;}$Mg+Cl2 | |
| D�� | ��ҵұ����Al2O3+2Fe$\frac{\underline{\;����\;}}{\;}$Fe2O3+2Al |
| A�� | �٢ۢ� | B�� | �ڢۢ� | C�� | �٢ڢ� | D�� | �ڢܢ� |
| A�� | Ư�ۺ�����������ˮ�����������߾�ˮԭ����ͬ | |
| B�� | ���ع��͡������ӹ������������Ʒ�������ʵ�ֳ������������� | |
| C�� | ̼������ҽ����������θ������һ��ҩ�� | |
| D�� | ��¯�����ij���FeS2��Ϊ������ԭ�� |
| A�� | 1mol FeCl3��ȫת��ΪFe��OH��3������γ�NA������ | |
| B�� | 1 L 0.1 mol/L Na2CO3��Һ��CO32-��HCO3-����Ϊ0.1 NA | |
| C�� | ��״���£�22.4L NO��11.2L O2��Ϻ�����ķ�������ΪNA | |
| D�� | 0.2 mol��Ȳ����ȩ�����������������ȫȼ�գ����������ķ�����Ϊ0.5NA |
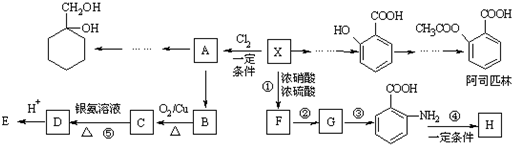
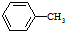 $\stackrel{KMnO_{4}/H+}{��}$
$\stackrel{KMnO_{4}/H+}{��}$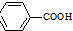
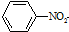 $\stackrel{Fe/HCl}{��}$
$\stackrel{Fe/HCl}{��}$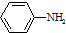 ���������ױ�������
���������ױ�������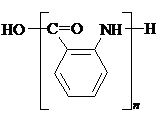 ��
��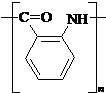 ��
��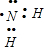 ��
��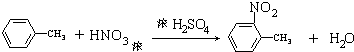 ��
��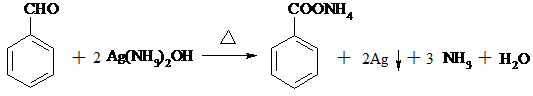 ��
��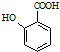 �ж���ͬ���칹�壬д��2�ֺ���1��ȩ����2���ǻ��ķ����廯����Ľṹ��ʽ��
�ж���ͬ���칹�壬д��2�ֺ���1��ȩ����2���ǻ��ķ����廯����Ľṹ��ʽ�� ��
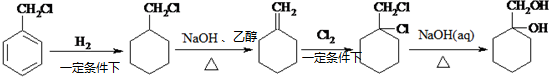
��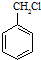 ����������ϳ�
����������ϳ� 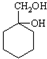 ������ķ�����������4������
������ķ�����������4������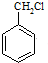 $��_{��Ӧ����}^{��Ӧ��}$��$��_{��Ӧ����}^{��Ӧ��}$
$��_{��Ӧ����}^{��Ӧ��}$��$��_{��Ӧ����}^{��Ӧ��}$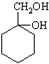
 ��
��