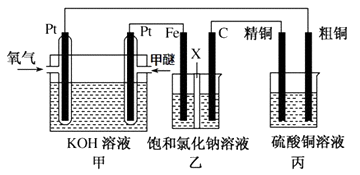
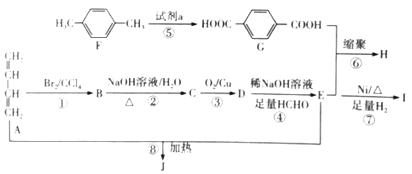
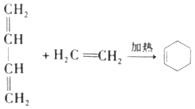
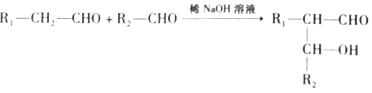��Ŀ����
����Ŀ����ˮ�ĵ���ƽ��������ͼ��ʾ��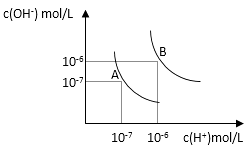
��1������A���ʾ25��ʱˮ�ĵ���ƽ�������Ũ�ȣ����¶����ߵ�100��ʱ��ˮ�ĵ���ƽ��״̬�ƶ���B�㣬���ʱˮ�����ӻ����仯����
��2����pH=8��Ba��OH��2��Һ��pH=5��ϡ�����ϣ�������100��ĺ��£���ʹ�����Һ��pH=7����Ba��OH��2������������Ϊ��
����A��B��C��D����ǿ����ʣ�������ˮ�е���ʱ�ɲ����������ӣ�ÿ������ֻ��һ���������һ����ظ����������� Na����Ba2����NH4+ ������ CH3COO����Cl����OH����SO42-����֪����A��C��Һ��pH������7��A��B����Һ��ˮ�ĵ���̶���ͬ����C��Һ��D��Һ����ʱֻ���ɰ�ɫ������B��Һ��C��Һ����ʱֻ���ɴ̼�����ζ�����壬A��Һ��D��Һ���ʱ����������
��A�� �� B�� ��
�������ӷ���ʽ��ʾA��Һ�ʼ��Ե�ԭ����
���𰸡�
��1��10-14,10-12
��2��2:9,CH3COONa,NH4Cl,CH3COO- + H2O ![]() CH3COOH + OH-
CH3COOH + OH-
�������� (1)ˮ�����ӻ�Kw=[H+][OH-]������ͼ��25�����ߵ�100��ʱ��ˮ�����ӻ���10-14�仯��10-12��
(2)����100��ĺ���, Kw=10-12��pH=8��Ba��OH��2��Һc��OH����= ![]() =10-4��pH=5��ϡ����c��H����=1��10��5�� pH=7����Һ�ʼ��ԣ�c��OH����=
=10-4��pH=5��ϡ����c��H����=1��10��5�� pH=7����Һ�ʼ��ԣ�c��OH����= ![]() =10-5������[OH-]=
=10-5������[OH-]= ![]() ,10-5=
,10-5= ![]() ,v(��)��v(��)=2:9��
,v(��)��v(��)=2:9��
�������Ϸ�������A��CH3COONa��B�� NH4Cl��
��CH3COONa��ǿ�������Σ����������ˮ�⣬��Һ�ʼ��ԣ����������ˮ��ķ���ʽ��CH3COO- + H2O ![]() CH3COOH + OH-��
CH3COOH + OH-��
�����㾫�����������ӻ���������Ŀ�����жϼ��ɵõ��𰸣���Ҫ��֪ˮ�����ӻ�Kw=c(H+)��c(OH�C)=10�C14��25�棩��Kwֻ���¶��йأ��¶����ߣ�Kw�����磺100�� Kw=10�C12��Kw�����ڴ�ˮ��ϡ�ᡢϡ�ϡ��ˮ��Һ����