��Ŀ����
��4�֣���֪̼�����Ƽ����ֽܷ�Ϊ̼���ơ�ˮ�Ͷ�����̼����̼�������Ȳ��ֽ⡣����ij������������һ��̼�������л���������̼���ƣ�ȡ4.0g ��Ʒ���ȵ��������ټ���ʱ�����ռ���������̼����0.88g��
��1��д��̼���������ȷֽ�Ļ�ѧ����ʽ ��
��2��Ҫ�õ�����Ķ�����̼���壬��ͼ��ʢ������ˮ��Ũ���ᣬ
���ɵ�����Ӧ�� ��ѡ�a����b�����˵��롣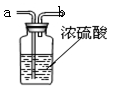
��3�������Ʒ��̼���Ƶ�������������д��������̣�
��1��2NaHCO3 Na2CO3��H2O��CO2��
Na2CO3��H2O��CO2��
��2��a ��3��16%
�������������
��1�����������֪��̼�����Ƽ����ֽܷ�Ϊ̼���ơ�ˮ�Ͷ�����̼����ѧ����ʽΪ��2NaHCO3��Na2CO3��H2O��CO2����
��2��Ҫ�ﵽ�����Ч�������ɵ�����Ӧ�ӳ��ܽ��룬�Ӷ̹��е���������Ӧ��a�˵��롣
��3�����������֪����֪��Ϊ������̼��������δ֪��Ϊ����Ʒ��̼���Ƶ�����������
����˼·���ɸ��ݶ�����̼��������������̼�����Ʒֽ�Ļ�ѧ����ʽ�������Ӧ��̼�����Ƶ���������һ�����δ֪�����������������£���д��������̣�
�⣺��̼�����Ƶ�����Ϊx��
2NaHCO3��Na2CO3��H2O��CO2��
168 44
x 0.88g
168��44=x��0.88g
x= 3.36g
����Ʒ��̼���Ƶ���������Ϊ��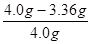 ��100%=16%
��100%=16%
�𣺸���Ʒ��̼���Ƶ���������Ϊ16%
���㣺��ѧ����ʽ����д������ľ������ۺϼ���

 ϰ�⾫ѡϵ�д�
ϰ�⾫ѡϵ�д�ijͬѧΪ�˲ⶨNaCl��MgCl2����������MgCl2��������������������ʵ�飺��80g������������ˮ�����Һ��ƽ���ֳ��ķݣ��ֱ������ͬ����������NaOH��Һ���������ʵ�����ݣ�
| ʵ����� | һ | �� | �� | �� |
| ���������������g�� | 20 | 20 | 20 | 20 |
| ����NaOH��Һ��������g�� | 20 | 40 | 60 | 80 |
| ���ɳ�����������g�� | 2��9 | m | 8��7 | 8��7 |
��2��ԭ����������MgCl2�����������Ƕ��٣���8�֣���Ҫ��д��������̣�
����̼���ơ�(2Na2CO3��3H2O2)�׳ƹ���˫��ˮ��������̼���ε����ʣ�����˫��ˮ�IJ��ȶ��Ժ������ԣ��Ǻܺõ����������������㷺Ӧ����ϴ�ӡ�ӡȾ��ҽҩ��������̼���ơ����Ʊ��������£�
��ش��������⣺
��1��H2O2�ڴ��������������ֽ⣬˵����ѧ�仯���ٶȺ� ��أ�
��2���ڡ���̼���ơ��м�����������ʱ�����ᵼ�¡���̼���ơ����ʵ��� ������ţ���
| A���������� | B��ϡ���� | C�������� | D��ʯ��ˮ |
2(2Na2CO3��3H2O2)����4Na2CO3��6H2O��3O2��
��ͨ������˵���������м���Ĺ��̣���
��������̼���ơ��ֽ��������12g����ͬʱ����̼���Ƶ������Ƕ��٣�
����������������̼����ȫ�����Ƴ�������������Ϊ10.6%����Һ������ˮ���ٺ�����
 CaO+CO2����
CaO+CO2����