��Ŀ����
11���ü�ʽ�廯þ����[��Ҫ�ɷ�ΪMg��OH��Br�������������л��ܼ����߷��ӻ�����]����C2H5Br��������ˮ���ܶ�Ϊ1.46g•cm-3����MgCl2•6H2O���ȿɼ�����Ⱦ�ֿɵû���ԭ�ϣ��йع���������ͼ��ʾ��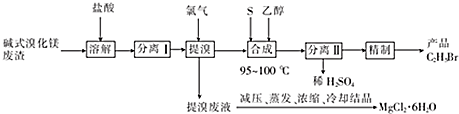
�ش��������⣺
��1����ʽ�廯þ��������ʱ��1molMg��OH��Br����0.5mol•L-1����������Ϊ2L��
��2�����������������˺ͷ�Һ����Ŀ���dz�ȥ���е��л���߷��ӻ������������ʱ����IJ�������Ϊ��Һ©�����ձ���
��3������ʱ����ҺԤ����60��ʱ��ʼͨ��������һ��ʱ�����Һ���¶Ȼ���Ȼ���ߵ�100�棬˵�����巴ӦΪ���ȣ�����ȡ������ȡ�����Ӧ�������Һ���ѹ��������Ũ����Ŀ����þ������ˮ�⣮
��4����Ũ������£��ϳ��������Ʒ�Ļ�ѧ����ʽΪ6CH3CH2OH+3Br2+S $\stackrel{Ũ����}{��}$6CH3CH2Br+H2SO4+2H2O���ϳ�ʱ����Ӧ�¶Ȳ��ܹ��ߣ�����Ҫԭ���Ǽ����Ҵ��Ļӷ�����ʧ��
��5������������IJ���Ϊˮϴ��Na2SO3��Һϴ�ӣ�ˮϴ���������Na2SO3��Һϴ�ӵ�Ŀ�������������ܽ���壮
��6���ö��Ե缫��һ�������µ��MgCl2��Һ����ȡ���������þ���õ�ⷴӦ�Ļ�ѧ����ʽΪMgCl2+6H2O$\frac{\underline{\;���\;}}{\;}$Mg��ClO3��2+6H2����
���� ��ʽ�廯þ����[��Ҫ�ɷ�ΪMg��OH��Br�������������л��ܼ����߷��ӻ�����]�������ܽ⣬ͨ����Һ��ȥ�л�����Ȼ�þ��Һ���廯þ��Һ�Ļ���ͨ�������û����壬��������Һ����Ҫ�����Ȼ�þ����������Ũ�������½ᾧ���Ȼ�þ���壬�������������Ҵ�������������������ᣬ������Һ������Ϊϡ���ᣬ�л��㺬�������飬���Ƶò�Ʒ�����飬�ݴ˴��⣮
��� �⣺��ʽ�廯þ����[��Ҫ�ɷ�ΪMg��OH��Br�������������л��ܼ����߷��ӻ�����]�������ܽ⣬ͨ����Һ��ȥ�л�����Ȼ�þ��Һ���廯þ��Һ�Ļ���ͨ�������û����壬��������Һ����Ҫ�����Ȼ�þ����������Ũ�������½ᾧ���Ȼ�þ���壬�������������Ҵ�������������������ᣬ������Һ������Ϊϡ���ᣬ�л��㺬�������飬���Ƶò�Ʒ�����飬
��1����ʽ�廯þ��������ʱ�����ݷ�Ӧ��2Mg��OH��Br+2HCl=MgBr2+MgCl2+2H2O������1molMg��OH��Br����1molHCl����0.5mol•L-1����������Ϊ2L��
�ʴ�Ϊ��2��
��2����������ķ�����֪�����������������˺ͷ�Һ����Ŀ���dz�ȥ���е��л���߷��ӻ������������Ϊ��Һ������ʱ����IJ�������Ϊ��Һ©�����ձ���
�ʴ�Ϊ����ȥ���е��л���߷��ӻ������Һ©�����ձ���
��3������ʱ����ҺԤ����60��ʱ��ʼͨ��������һ��ʱ�����Һ���¶Ȼ���Ȼ���ߵ�100�棬˵�����巴ӦΪ���ȷ�Ӧ����Ϊþ������ˮ�⣬���������Һ���ѹ��������Ũ�����Ȼ�þ���壬
�ʴ�Ϊ�����ȣ�þ������ˮ�⣻
��4���塢���Ҵ���Ũ������·�Ӧ���������飬��Ӧ�Ļ�ѧ����ʽΪ6CH3CH2OH+3Br2+S $\stackrel{Ũ����}{��}$6CH3CH2Br+H2SO4+2H2O���ϳ�ʱ����Ӧ�¶Ȳ��ܹ��ߣ�����Ҫԭ���Ǽ����Ҵ��Ļӷ�����ʧ��
�ʴ�Ϊ��6CH3CH2OH+3Br2+S $\stackrel{Ũ����}{��}$6CH3CH2Br+H2SO4+2H2O�������Ҵ��Ļӷ�����ʧ��
��5�����������к����壬�����ھ��ƹ�������Na2SO3��Һϴ�ӵ�Ŀ���dz�ȥ���������ܽ���壬
�ʴ�Ϊ�����������ܽ���壻
��6���ö��Ե缫��һ�������µ��MgCl2��Һ�������������ӵõ�������������������������ʧ�����������������ⷴӦ�Ļ�ѧ����ʽΪMgCl2+6H2O$\frac{\underline{\;���\;}}{\;}$Mg��ClO3��2+6H2����
�ʴ�Ϊ��MgCl2+6H2O$\frac{\underline{\;���\;}}{\;}$Mg��ClO3��2+6H2����
���� �������Ȼ�þ����������Ʊ�ʵ�鷽��Ϊ���壬����Ӧ�û���֪ʶ�����ѧ����������Լ���ͼ���Ĺ۲졢�������������̷���Ӧ�ã����������ķ������������ӵij���ԭ����Լ�ѡ����Ŀ�Ѷ��еȣ�


| A�� | ��ͼ1��ʾװ�ô�KI��I2�Ĺ��������л���I2 | |
| B�� | ��ͼ2��ʾװ������100mL0.10mol•L-1 K2Cr2O7��Һ | |
| C�� | ��ͼ3��ʾװ�ø���������SO2������ | |
| D�� | ��ͼ4��ʾװ������FeCl3������Һ�Ʊ�FeCl3���� |
| ѡ�� | ʵ�� | ���� | ���� |
| A | ��Ũ������������� | �����ں������� | Ũ���������ˮ�Ժ�ǿ��ԭ�� |
| B | ��������ͬ���Ʒֱ�Ͷ���Ҵ���ˮ�� | ������ˮ�з�Ӧ�����Ҵ��о��� | ��ͬ�����£�ˮ�ĵ����������Ҵ�ǿ |
| C | ��ij��Һ�еμ���ˮ���ٵ���KSCN��Һ | ��Һ��Ϊ��ɫ��Һ��Ϊ��ɫ | ����Һ��һ������Fe3+ |
| D | ����ʱ����Ƭ����Ũ������ | δ�������� | ����������Ũ�����Ӧ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ƽ�����淴Ӧ����ת�� | B�� | ƽ�ⲻ�ƶ� | ||
| C�� | C�İٷֺ������� | D�� | C�İٷֺ�����С |
| A�� | NH3+H3O+�TNH${\;}_{4}^{+}$+H2O��NH3���H+��������H2Oǿ | |
| B�� | Al2O3+2NaOH�T2NaAlO2+H2O��Al2O3������������ | |
| C�� | HClO+SO2+H2O�THCl+H2SO4������ HClO��H2SO4 | |
| D�� | ��֪C��s��ʯī��=C��s�����ʯ����H=+1.9 kJ/mol�����ʯ��ʯī�ȶ� |
| A�� | �߲˱����������������֣��ɱ��ʺ����� | |
| B�� | �ƹ�ʹ�ÿɽ������ϼ����ʹ�������Լ��١���ɫ��Ⱦ�� | |
| C�� | ��ɫʳƷ���Dz�ʹ�û���ũҩ�������κλ�ѧ���ʵ�ʳƷ | |
| D�� | �ƹ�ʹ��úҺ�������ɼ��ٶ�����̼����������ŷ� |
 ���Ƴɵ�������������������ڿɽ��⣬ʡȥ�˷��Ƥ������ߵ��鷳���ϳ�PLGA��һ��·����ͼ��
���Ƴɵ�������������������ڿɽ��⣬ʡȥ�˷��Ƥ������ߵ��鷳���ϳ�PLGA��һ��·����ͼ��

 ��
�� ��
�� �ĺϳ���·��
�ĺϳ���·�� ��
�� ij�о���ѧϰС����̽���ں�Mg2+��Al3+�Ļ����Һ�еμ�NaOH��Һʱ������������������Ĺ��̣�
ij�о���ѧϰС����̽���ں�Mg2+��Al3+�Ļ����Һ�еμ�NaOH��Һʱ������������������Ĺ��̣�