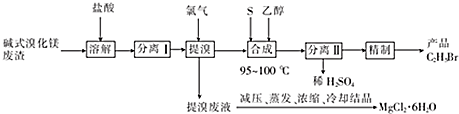
��Ŀ����
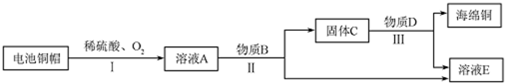
8��ijʵ��С��������ͼװ�ý����Ҵ��Ĵ�����ʵ�飬����ȡ��ȩ����װ������ˮ���ղ��������̨�ȼг�����ʡ�ԣ�ʵ��ʱ���ȼ��Ȳ������е�ͭ˿��Լ1min����������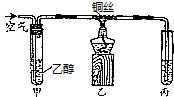
��1���Ҵ������������Ļ�ѧ����ʽΪ2CH3CH2OH+O2$��_{��}^{Cu}$2CH3CHO+2H2O��
��2��ʵ��ʱ��������װ�ý���70�桫80���ˮԡ�м��ȣ���װ�õ��������ʵ��ӿ������Ҵ����������ʣ����ƽ�ȵ��Ҵ���������2������
��3�����ڱ�װ������ϵ�ȱ�ݣ�ʵ�����ʱ���ܻᷢ��������
��4����ͭ˿���ɹ۲쵽���Ȳ��ֵ�ͭ˿���Ъ�Եع��˿�����������ֱ�ڱ�����ʵ������
��5������֤����Ӧ��������ȩ���������д̼�����ζ������
���� ��װ�ã�ʵ��ʱ���ȼ��Ȳ������е�ͭ˿��Լ1min�������������ƽ�ȵ��Ҵ��Ϳ�������������װ�ã��Ҵ�����������������ȩ��ˮ������ʽΪ2CH3CH2OH+O2$��_{��}^{Cu}$2CH3CHO+H2O����װ�ã����ղ��
��1���Ҵ���ͭ�������±�����������ȩ��
��2���Ҵ����лӷ��ԣ������¶��ܴٽ��Ҵ��Ļӷ�������ʱ����ˮԡ���ȿ������Ҵ�ƽ���������Ҵ�������
��3�����ɵ���ȩ��δ��Ӧ���Ҵ�������ˮ��
��4��ͭ��������Ӧ���ɺ�ɫ������ͭ������ͭ���Ҵ���Ӧ����ԭΪͭ��
��5����ȩ���д̼�����ζ���ӷ���
��� �⣺��1���Ҵ���������������ȩ��ˮ����Ӧ����ʽΪ��2CH3CH2OH+O2$��_{��}^{Cu}$2CH3CHO+2H2O���ʴ�Ϊ��2CH3CH2OH+O2$��_{��}^{Cu}$2CH3CHO+2H2O��
��2����װ�ó��������¶�Ϊ70��80��ˮԡ�У�ˮԡ��ʹ�������Ⱦ��ȣ���ʹ�����Ҵ�ƽ���������Ҵ�������
�ʴ�Ϊ���ʵ��ӿ������Ҵ����������ʣ����ƽ�ȵ��Ҵ�������
��3��������ֱ�Ӳ������Һ�ڣ����ɵ���ȩ�������Ҵ�������ˮ��ʹ��װ�ò�����������
�ʴ�Ϊ������������
��4������ʱ��Cu����������CuO��Ȼ��CuO�����Ҵ�������ȩ��Cu��ˮ���ظ�����������Ӧ����ɹ۲쵽A�����Ȳ��ֵ�ͭ˿���Ъ�Եع��˿�����������ֱ�ڱ�����
�ʴ�Ϊ�����Ȳ��ֵ�ͭ˿���Ъ�Եع��˿�����������ֱ�ڱ�����
��5����ȩ��һ�־��д̼�����ζ���ӷ���Һ�壬������г��ִ̼�����ζ���Ϳ�֤������ȩ���ɣ��ʴ�Ϊ���д̼�����ζ������
���� ���⿼�����Ҵ��Ĵ�����ʵ�飬�����Ҵ��Ļ�ѧ�����Լ��Ҵ��Ĵ�����ʵ�����ʱ���Ĺؼ�����Ŀ�Ѷ��еȣ�

 ��У����ϵ�д�
��У����ϵ�д�| A�� | ������ӵ����ģ�ͣ�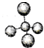 | B�� | NH4Br�ĵ���ʽ��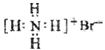 | ||
| C�� | ��ԭ�ӵĽṹʾ��ͼ�� | D�� | ���ǻ�������Ľṹ��ʽ��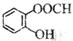 |
| A�� | ������Ϊ�л��� | B�� | ������Ϊ���� | ||
| C�� | ������������Ԫ����� | D�� | ������ȼ�ղ�����ܺ�CO2 |
| A�� | Cu | B�� | CaCO3 | C�� | Zn | D�� | AgNO3 |
| A�� | ��������������ˮ�У���ʹ��̪��� | |
| B�� | �������ֺ��� | |
| C�� | �������ɺ���ɫ������ | |
| D�� | ���ܲ������� |