��Ŀ����
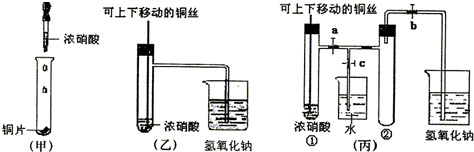
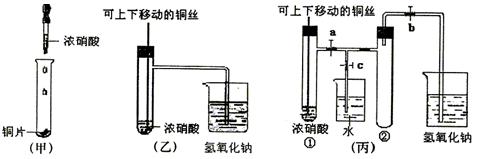
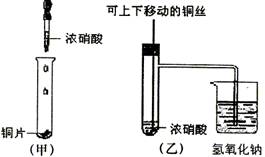
��ͼ���ǿα�����֤ͭ��Ũ���ᷴӦ��װ�ã��ҡ�����ʦ������ʾʵ��Ľ����װ�ã�
(1)д��ͭ��Ũ���ᷴӦ�Ļ�ѧ����ʽ��
____________________________________________________________________��
(2)�ͼ�װ����ȣ���װ�õ��ŵ���
____________________________________________________________________��
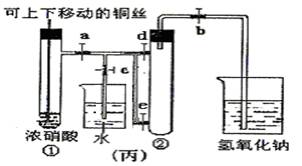
(3)Ϊ�˽�һ����֤NO2��ˮ�ķ�Ӧ��ijѧ������˱�װ�ã���ʵ��ʱ�ȹرյ��ɼ�___________���ٴ��ɼ�___________������ʹNO2����������Թܡ�
(4)������������Թܺ�ͭ˿��������Һ���룬��ʹ�ձ��е�ˮ������Թ�Ӧ��β�����______________________��
(5)���Թ��е�NO2��ˮ��ַ�Ӧ��������Һ���ʵ���Ũ�ȵ����ֵ��___________(�����������״������)��
(1)Cu+4HNO3(Ũ)![]() Cu(NO3)2+2NO2��+2H2O
Cu(NO3)2+2NO2��+2H2O
(2)�ٿ��Կ��Ʒ�Ӧ��������NO2���壬��ֹ��Ⱦ����
(3)c a��b
(4)�ȹر�b���ٹر�a��Ȼ���c��������ס(��ˮ����ë��������)�Թܢ�(�����𰸾���)
(5)1/22.4 mol��L-1��0.045 mol��L-1
������(2)�ҿ���ͨ�����³鶯ͭ˿������HNO3�ķ�Ӧ���ʣ��Ҳ���NO2�ɱ�NaOH����.
(4)�У�ʹ�ձ��е�H2O������Թ��еIJ��������Խ����Ȫʵ��IJ���ԭ�������������������ܷ⣬���ȼ���.

 ȫ�ܲ��һ���þ�ϵ�д�
ȫ�ܲ��һ���þ�ϵ�д�