��Ŀ����
ͼΪԪ�����ڱ���һ���֣������Ԫ�آ�-����ͼ�е�λ�ã��û�ѧ����ش��������⣺

��1������ʲôԪ�� ����Ԫ�ط��ű�ʾ����
��2���뻭�����ԭ�ӽṹʾ��ͼ ��
��3���ڡ��ۡ��ܡ��ݵİ뾶�Ӵ�С��˳���� ����Ԫ�ط��ű�ʾ����
��4�����������Ԫ�ؽ�����ǿ��˳��Ϊ���� �ߣ������������=����
��5��������γɵĻ�����ĵ���ʽ��
��6����д����Ԫ�����������Ļ�ѧʽ�� ��������������Ӧ��ˮ������ߵ�����������Ӧ��ˮ�������Ӧ�Ļ�ѧ����ʽ�� ��

��1������ʲôԪ��
��2���뻭�����ԭ�ӽṹʾ��ͼ
��3���ڡ��ۡ��ܡ��ݵİ뾶�Ӵ�С��˳����
��4�����������Ԫ�ؽ�����ǿ��˳��Ϊ����
��5��������γɵĻ�����ĵ���ʽ��
��6����д����Ԫ�����������Ļ�ѧʽ��
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
����������Ԫ�������ڱ��е�λ��֪���٢ڢۢܢݢޢߢ��ֱ���H��C��N��O��F��Na��Al��S��ClԪ�أ�
��1��������Ԫ�أ�
��2����ԭ�Ӻ�����3�����Ӳ㡢17�����ӣ�
��3��ͬһ����Ԫ�أ�ԭ�Ӱ뾶����ԭ�������������С��
��4��ͬһ����Ԫ�أ�Ԫ�صĽ���������ԭ�����������������
��5��������γɵĻ�����Ϊˮ�����ڹ��ۻ����
��6����Ԫ�����������Ϊ������������������Ӧ��ˮ������NaOH���ߵ�����������Ӧ��ˮ������Al��OH��3�����߷�Ӧ����ƫ�����ƺ�ˮ��
��1��������Ԫ�أ�
��2����ԭ�Ӻ�����3�����Ӳ㡢17�����ӣ�
��3��ͬһ����Ԫ�أ�ԭ�Ӱ뾶����ԭ�������������С��
��4��ͬһ����Ԫ�أ�Ԫ�صĽ���������ԭ�����������������
��5��������γɵĻ�����Ϊˮ�����ڹ��ۻ����
��6����Ԫ�����������Ϊ������������������Ӧ��ˮ������NaOH���ߵ�����������Ӧ��ˮ������Al��OH��3�����߷�Ӧ����ƫ�����ƺ�ˮ��
���
����Ԫ�������ڱ��е�λ��֪���٢ڢۢܢݢޢߢ��ֱ���H��C��N��O��F��Na��Al��S��ClԪ�أ�
��1��������Ԫ�أ���Ԫ�ط���ΪAl���ʴ�Ϊ��Al��
��2����ԭ�Ӻ�����3�����Ӳ㡢17�����ӣ���ԭ�ӽṹʾ��ͼΪ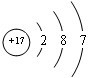 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3��ͬһ����Ԫ�أ�ԭ�Ӱ뾶����ԭ�������������С������ԭ�Ӱ뾶��С˳���Ǣڣ��ۣ��ܣ��ݣ��ʴ�Ϊ���ڣ��ۣ��ܣ��ݣ�
��4��ͬһ����Ԫ�أ�Ԫ�صĽ���������ԭ����������������������Ԣޣ��ߣ��ʴ�Ϊ������
��5��������γɵĻ�����Ϊˮ�����ڹ��ۻ��������ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��6����Ԫ�����������Ϊ��������ѧʽΪSO3��������������Ӧ��ˮ������NaOH���ߵ�����������Ӧ��ˮ������Al��OH��3�����߷�Ӧ����ƫ�����ƺ�ˮ����Ӧ����ʽΪAl��OH��3+NaOH=NaAlO2+2H2O���ʴ�Ϊ��SO3��Al��OH��3+NaOH=NaAlO2+2H2O��
��1��������Ԫ�أ���Ԫ�ط���ΪAl���ʴ�Ϊ��Al��
��2����ԭ�Ӻ�����3�����Ӳ㡢17�����ӣ���ԭ�ӽṹʾ��ͼΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����3��ͬһ����Ԫ�أ�ԭ�Ӱ뾶����ԭ�������������С������ԭ�Ӱ뾶��С˳���Ǣڣ��ۣ��ܣ��ݣ��ʴ�Ϊ���ڣ��ۣ��ܣ��ݣ�
��4��ͬһ����Ԫ�أ�Ԫ�صĽ���������ԭ����������������������Ԣޣ��ߣ��ʴ�Ϊ������
��5��������γɵĻ�����Ϊˮ�����ڹ��ۻ��������ʽΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����6����Ԫ�����������Ϊ��������ѧʽΪSO3��������������Ӧ��ˮ������NaOH���ߵ�����������Ӧ��ˮ������Al��OH��3�����߷�Ӧ����ƫ�����ƺ�ˮ����Ӧ����ʽΪAl��OH��3+NaOH=NaAlO2+2H2O���ʴ�Ϊ��SO3��Al��OH��3+NaOH=NaAlO2+2H2O��
���������⿼����Ԫ�������ɺ�Ԫ�����ڱ��ۺ�Ӧ�ã�Ԫ�������ڱ�λ��ȷ��Ԫ�أ��ٽ��Ԫ�������ɷ��������Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
 �����в����еĻ����ǣ�������
�����в����еĻ����ǣ�������| A���� | B���ǻ� | C���Ȼ� | D��ȩ�� |
Ӧ��Ԫ�������ɺ�Ԫ�����ڱ����ж�����˵����ȷ���ǣ�������
| A����������ʵ��۵���ԭ����������������� |
| B������At���Ǣ�A��Ԫ�أ�HAt���ȶ��Դ���HCl |
| C����2���ڷǽ�������̬�⻯������ˮ��������Һ��Ϊ���� |
| D���裨Tl������ͬ���壬�䵥�ʼ��������ᷴӦ������NaOH��Һ��Ӧ |