��Ŀ����
ˮ��Һ�д��ڶ���ƽ�⣬�������ѧ��ѧ֪ʶ�ش��������⣺
��1����Ũ�Ⱦ�Ϊ0.1mol/L��CH3COONH4 ��NH4HSO4 ��NH3.H2O ��(NH4)2SO4��Һ�У�NH4+Ũ���ɴ�С��˳��Ϊ ������ţ�
��2��Ũ�Ⱦ�Ϊ0.1 mol��L��1��������Һ�������ᡢ�ڴ��ᡢ���������ơ����Ȼ��������Һ����ˮ�������H��Ũ���ɴ�С��˳����(�����) ��
��3�������£���pH=6������ˮ�м���2.3g�����ƣ���ַ�Ӧ���ټ�����ˮϡ�͵�1L��������Һ��pH= ��
��1����Ũ�Ⱦ�Ϊ0.1mol/L��CH3COONH4 ��NH4HSO4 ��NH3.H2O ��(NH4)2SO4��Һ�У�NH4+Ũ���ɴ�С��˳��Ϊ ������ţ�
��2��Ũ�Ⱦ�Ϊ0.1 mol��L��1��������Һ�������ᡢ�ڴ��ᡢ���������ơ����Ȼ��������Һ����ˮ�������H��Ũ���ɴ�С��˳����(�����) ��
��3�������£���pH=6������ˮ�м���2.3g�����ƣ���ַ�Ӧ���ټ�����ˮϡ�͵�1L��������Һ��pH= ��
��1����>��>��>�ۣ�2����>��>��>�٣�3��11
�����������1����Ũ�Ⱦ�Ϊ0.1mol/L��CH3COONH4 ��NH4HSO4 ��NH3.H2O ��(NH4)2SO4��Һ�У�NH3?H2OΪ������ʣ����������ģ���CH3COONH4��NH4HSO4 ��(NH4)2SO4Ϊǿ����ʣ�ȫ�����룬����NH3.H2O��NH4+Ũ����С��(NH4)2SO4��������NH4+����(NH4)2SO4��NH4+Ũ�����NH4HSO4������������ӣ�NH4+ˮ���ܵ����ƣ�CH3COONH4�д����ˮ��ʼ��ԣ�NH4+ˮ��õ��ٽ�����NH4HSO4��NH4+Ũ�ȴ���CH3COONH4��NH4+Ũ�ȣ���NH4+Ũ���ɴ�С��˳��Ϊ ��>��>��>�ۣ���2�� Ũ�Ⱦ�Ϊ0.1 mol��L��1��������Һ�������ᡢ�ڴ��ᡢ���������ơ����Ȼ��������Һ���Ȼ��Ϊǿ�������Σ�����ˮ�⣬�ٽ�ˮ�ĵ��룬��ˮ�������H��Ũ��������ᡢ��������ˮ�ĵ��룬����Һ�Ƚ�ˮ��������������ӣ��ɱȽ���Һ����������Ũ�ȣ�����Ϊǿ�ᣬȫ�����룬0.1 mol��L��1��������������Ũ��Ϊ0.2mol��L��1��������Ũ��Ϊ5��10��14mol��L��1������Ϊ���ᣬ0.1 mol��L��1������Һ��������Ũ��ԶС��0.1 mol��L��1��������Ũ��Զ����1��10��13mol��L��1��������������ˮ�ĵ��룬����Һ�Ƚ�ˮ��������������ӣ��ɱȽ���Һ�������ӵ�Ũ�ȣ�0.1 mol��L��1����������Һ��������Ũ��Ϊ0.1 mol��L��1����Һ��������Ũ��Ϊ1��10��13mol��L��1������������������Һ����ˮ�������H��Ũ���ɴ�С��˳���Ǣ�>��>��>�٣���3�������£���pH=6������ˮ�м���2.3g�����ƣ�������Ӧ2Na+2H2O=2NaOH+H2��������0.1mol�������ƣ��ټ�����ˮϡ�͵�1L��������Һ��������Ũ��Ϊ0.1 mol��L��1�����¶�������ˮ��pH=6����ˮ�����ӻ�����Ϊ1��10��12������Һ��������Ũ��Ϊ1��10��11mol��L��1��pH=11��

��ϰ��ϵ�д�
 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�
�����Ŀ
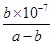 mol��L-1
mol��L-1 B++OH-
B++OH- NH4+ +OH-�������м�������0.01 mol��L-1NaOH��Һ�����ֲ������( )
NH4+ +OH-�������м�������0.01 mol��L-1NaOH��Һ�����ֲ������( )