��Ŀ����
13�������Ǻϳɷ������л��߾���PC�ĺϳ�·�ߣ�
��֪��ROH+R��OH$��_{��}^{Ũ����}$ ROR��+H2O
���������գ�
��1��F�й����ŵ������Ȼ�����ԭ�ӣ�д����Ӧ�ٵķ�Ӧ�������գ�
��2��д����Ӧ�ݵĻ�ѧ����ʽHOOCCCl��CH3��CH2COOH+3NaOH$��_{��}^{�Ҵ�}$
 +NaCl+3H2O��
+NaCl+3H2O����3��д���߾���P�Ľṹ��ʽ
 ��
����4��E�ж���ͬ���칹�壬д��һ�ַ�������������ͬ���칹��Ľṹ��ʽ
 ��
���ٷ�����ֻ�б���һ����״�ṹ���ұ�����������ȡ������
��1mol���л�������ˮ��Ӧʱ����4molBr2
��5��д���Է���ʽΪC5H8����Ϊ��Ҫԭ�ϣ��Ʊ�F�ĺϳ�·������ͼ�����Լ���ѡ�����ϳ�·������ͼʾ�����£�CH3CHO$��_{����}^{O_{2}}$ CH3COOH$��_{ŨH_{2}SO_{4}}^{CH_{3}CH_{2}OH}$ CH3COOCH2CH3��
���� ��C7H8������Ӧ�����ɵ�C7H7Cl�ܹ���NaOH��Һ�з�Ӧ��֪��C7H8Ϊ�ױ����ױ��ڹ��������£����ϵ���ԭ�ӱ���ԭ��ȡ����C7H7ClΪ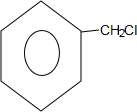 ��CΪ���״��������֪��Ӧ�ͷ�Ӧ������֪��DΪ
��CΪ���״��������֪��Ӧ�ͷ�Ӧ������֪��DΪ ��D��E����Ũ���������´�����ȥ��Ӧ����EΪ
��D��E����Ũ���������´�����ȥ��Ӧ����EΪ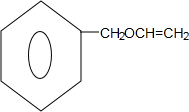 ����M�ķ���ʽ��֪��F��M��������ȥ��Ӧ���кͷ�Ӧ��MΪ
����M�ķ���ʽ��֪��F��M��������ȥ��Ӧ���кͷ�Ӧ��MΪ ��NΪ
��NΪ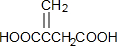 ��E��N�����Ӿ۷�Ӧ�õ��߾���P��PΪ
��E��N�����Ӿ۷�Ӧ�õ��߾���P��PΪ ���ݴ˷�����
���ݴ˷�����
��� �⣺��1����F�Ľṹ��ʽ��֪��F�к��еĹ��������Ȼ�����ԭ�ӣ���Ӧ�ٵķ�Ӧ����Ϊ���գ��ʴ�Ϊ���Ȼ�����ԭ�ӣ����գ�
��2��F��M��������ȥ��Ӧ���кͷ�Ӧ��MΪ ����Ӧ�Ļ�ѧ����ʽΪ��HOOCCCl��CH3��CH2COOH+3NaOH$��_{��}^{�Ҵ�}$
����Ӧ�Ļ�ѧ����ʽΪ��HOOCCCl��CH3��CH2COOH+3NaOH$��_{��}^{�Ҵ�}$ +NaCl+3H2O���ʴ�Ϊ��HOOCCCl��CH3��CH2COOH+3NaOH$��_{��}^{�Ҵ�}$
+NaCl+3H2O���ʴ�Ϊ��HOOCCCl��CH3��CH2COOH+3NaOH$��_{��}^{�Ҵ�}$ +NaCl+3H2O��
+NaCl+3H2O��
��3��EΪ ��NΪ
��NΪ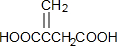 ��E��N�����Ӿ۷�Ӧ�õ��߾���P��PΪ
��E��N�����Ӿ۷�Ӧ�õ��߾���P��PΪ ��
��
�ʴ�Ϊ�� ��
��
��4��EΪ ����ͬ���칹����Тٷ�����ֻ�б���һ����״�ṹ���ұ�����������ȡ��������1mol���л�������ˮ��Ӧʱ����4molBr2�����ʵĽṹ��ʽ������
����ͬ���칹����Тٷ�����ֻ�б���һ����״�ṹ���ұ�����������ȡ��������1mol���л�������ˮ��Ӧʱ����4molBr2�����ʵĽṹ��ʽ������ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��5���÷���ʽΪC5H8����Ϊ��Ҫԭ�ϣ��Ʊ�F�ĺϳ�·������ͼΪCH2=C��CH3��CH=CH2$\stackrel{��ˮ}{��}$CH2BrC��CH3��=CHCH2Br$��_{��}^{NaOH��Һ}$HOCH2C��CH3��=CHCH2OH$\stackrel{������}{��}$OHCC��CH3��=CHCHO$\stackrel{������}{��}$HOOCC��CH3��=CHCOOH$��_{����}^{HCl}$F��
�ʴ�Ϊ��CH2=C��CH3��CH=CH2$\stackrel{��ˮ}{��}$CH2BrC��CH3��=CHCH2Br$��_{��}^{NaOH��Һ}$HOCH2C��CH3��=CHCH2OH$\stackrel{������}{��}$OHCC��CH3��=CHCHO$\stackrel{������}{��}$HOOCC��CH3��=CHCOOH$��_{����}^{HCl}$F��
���� ���⿼�����л��ƶ���ϳɣ�ע������л���Ĺ����źͷ�Ӧ��������������Ĺؼ��Ǿ�F�Ľṹ��ʽ��D�ķ���ʽ�����жϣ���Ŀ�ѶȽϴ�

 ��У����ϵ�д�
��У����ϵ�д�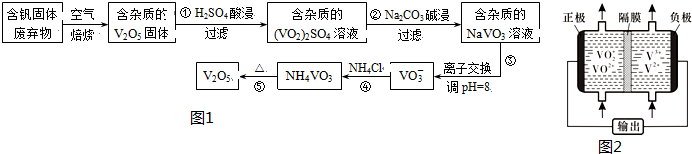
���ֺ�����������ˮ�е��ܽ��������
| ���� | V2O5 | NH4VO3 | VOSO4 | ��VO2��2SO4 |
| �ܽ��� | ���� | ���� | ���� | ���� |
��1����Ӧ��������Һ�г�H+֮�����������VO2+��Al3+
��2����Ӧ�ڼ�����˳��Ĺ�����Ҫ�ɷ���Al��OH��3��д��ѧʽ����
��3����Ӧ�ܵ����ӷ���ʽΪVO3-+NH4+=NH4VO3����
��4��25�桢101 kPaʱ��4Al��s��+3O2��g���T2Al2O3��s����H1=-a kJ/mol
4V��s��+5O2��g���T2V2O5��s����H2=-b kJ/mol
��V2O5�������ȷ�Ӧұ�����������Ȼ�ѧ����ʽ��10Al��s��+3V2O5��s��=5Al2O3��s��+6V��s����H=-$\frac{5a-3b}{2}$KJ/mol��
��5����Һ����أ���ͼ2��ʾ�����й�����Ӧ��������г�ǰ�����õ���и�Ĥֻ����H+ͨ������طŵ�ʱ�����ĵ缫��ӦʽΪV2+-e-=V3+����س��ʱ�����ĵ缫��Ӧʽ��VO2+-e-+H2O=VO2++2H+��
��6���������ữ��H2C2O4��Һ�ζ���VO2��2SO4��Һ���Բⶨ��Ӧ�ٺ���Һ�еĺ���������Ӧ�����ӷ���ʽΪ��2VO+H2C2O4+2H+�T2VO2++2CO2��+2H2O��ȡ25.00mL 0.1000 mol/LH2C2O4����Һ����ƿ�У�����ָʾ����������Һʢ���ڵζ����У��ζ����յ�ʱ���Ĵ���Һ24.0mL���ɴ˿�֪���ã�VO2��2SO4��Һ�з��ĺ���Ϊ10.6g/L��
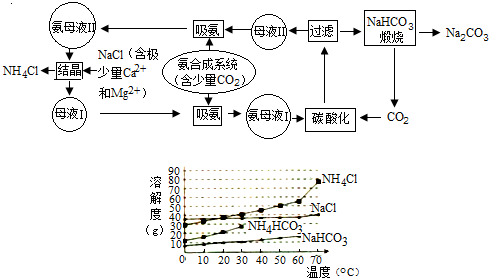
| �¶�/�� | 10 | 20 | 30 | 40 | 50 | 70 |
| ��NH4��2SO4 | 73.0 | 75.4 | 78.0 | 81.0 | 84.5 | 91.9 |
| FeSO4•7H2O | 40.0 | 48.0 | 60.0 | 73.3 | - | - |
| Ħ���� | 18.1 | 21.2 | 24.5 | 27.9 | 31.3 | 38.5 |
�ش��������⣺
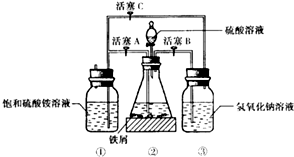
��1������30%������������Һ��з���м�����������ۡ����⡢FeS�ȣ���������ˮϴ����������������Һ��е�Ŀ���dz�ȥ��м������
��2���������õ���м������ƿ�У�����ϡ���ᣮ��ƿ�з�����Ӧ�����ӷ���ʽ����ΪABCD������ţ���
A��Fe+2H+�TFe2++H2��
B��Fe2O3+6H+�T2Fe3++3H2O
C��2Fe3++S2-�T2Fe2++S��
D��2Fe3++Fe�T3Fe2+
��3�����������ڵķ�Ӧ������������ͨ��������Ӧ�رջ���A������BC ������ĸ������������NaOH��Һ�������������������壬��ֹ��Ⱦ����������������ͨ��������Ŀ���Ƿ�ֹ�������ӱ�����������
����ƿ�е���м�췴Ӧ��ʱ���رջ���B��C������A�����������������Ὣ��ƿ�е����������������ٲ���δ��Ӧ��ϡ���ᣩѹ�������������Һ�ĵײ����ڳ����·���һ��ʱ�䣬�Լ�ƿ�ײ����ᾧ����������泥�
�����������������Һ��Ͼ��ܵõ���������茶��壬��ԭ������������淋��ܽ����С�����������з��벢�õ�������������茶���IJ��������ǹ��ˡ��þƾ�ϴ�ӡ����
���Ƶõ���������茶������������м�������Fe3+��Ϊ�ⶨ������Fe2+�ĺ�������ȡһ������Ϊ20.0g����������茶�����Ʒ���Ƴ���Һ����0.5mo1•L-1KMnO4��Һ�ζ�������Һ��Fe2+ȫ����������MnO-4����ԭ��Mn2+ʱ����KMnO4��Һ���20.00mL��
�ζ�ʱ����KMnO4��Һװ����ʽ����ʽ���ʽ���ζ����У��жϷ�Ӧ����ζ��յ������Ϊ��Һ�ճ����Ϻ�ɫ������30s���䣻������FeSO4����������Ϊ38%��
| A�� | ����ʱ��̫�� | B�� | ��Ӧ��ȴ���ٵ���AgNO3��Һ | ||
| C�� | ��AgNO3��Һǰδ��ϡHNO3�ữ | D�� | ��AgNO3��Һ��δ��ϡHNO3 |
 ��ѧ��Ӧ����Ϊ�ɼ����Ѻ��¼��γɵĹ��̣���ѧ���ļ������γɣ����1mol��ѧ��ʱ�ͷţ������գ�����������֪����P4O6�ķ��ӽṹ����ͼ��ʾ�����ṩ���»�ѧ���ļ��ܣ�kJ/mol����P-P��198��P-O��360��O=O��498����ӦP4�����ף�+3O2�TP4O6�ķ�Ӧ�ȡ�HΪ��������
��ѧ��Ӧ����Ϊ�ɼ����Ѻ��¼��γɵĹ��̣���ѧ���ļ������γɣ����1mol��ѧ��ʱ�ͷţ������գ�����������֪����P4O6�ķ��ӽṹ����ͼ��ʾ�����ṩ���»�ѧ���ļ��ܣ�kJ/mol����P-P��198��P-O��360��O=O��498����ӦP4�����ף�+3O2�TP4O6�ķ�Ӧ�ȡ�HΪ��������| A�� | -1 638 kJ/mol | B�� | +1 638 kJ/mol | C�� | -126 kJ/mol | D�� | +126 kJ/mol |

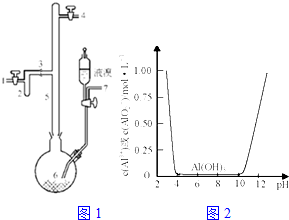 ���÷���������Ҫ�ɷ�ΪAl��������Fe��Si�ȣ��ȿ���ȡ�л��ϳɴ���AlBr3�ֿ���ȡ��ˮ�����������壮
���÷���������Ҫ�ɷ�ΪAl��������Fe��Si�ȣ��ȿ���ȡ�л��ϳɴ���AlBr3�ֿ���ȡ��ˮ�����������壮